Abstract
Objective: Optic nerve sheath diameter (ONSD) has been increasingly recognized as a non-invasive surrogate marker for intracranial pressure (ICP) in critically ill patients. The Glasgow Coma Scale (GCS) is a widely used tool for assessing neurological status. However, the relationship between GCS and ONSD in intensive care unit (ICU) patients remains underexplored. This study aimed to investigate the association between ONSD and GCS scores in ICU patients, while also evaluating the impact of positive end-expiratory pressure (PEEP) on ONSD measurements.
Methods: This prospective observational study included 66 ICU patients. ONSD measurements were obtained using ultrasonography at 24 and 48–72 hours after ICU admission. Patients were categorized into three groups based on GCS scores (3–7, 8–11, 12–15). The effect of PEEP and other clinical parameters on ONSD was analyzed using linear mixed models, with a significance level of p < 0.05.
Results: ONSD was significantly higher in patients with lower GCS scores (p < 0.001). Additionally, PEEP application was strongly associated with increased ONSD (p < 0.001), whereas other factors such as mean arterial pressure, heart rate, and sepsis status did not show significant effects.
Conclusion: ONSD is significantly associated with GCS scores, supporting its role as a non-invasive marker of neurological deterioration. The influence of PEEP on ONSD suggests a potential impact of mechanical ventilation on ICP dynamics, warranting further investigation.
Keywords: optic nerve sheath diameter, Glasgow Coma Scale, intracranial pressure, ultrasonography, intensive care unit
Introduction
Optic nerve sheath diameter (ONSD) has emerged as a promising non-invasive indicator of increased intracranial pressure (ICP) in critically ill patients. Ultrasonographic assessment of ONSD allows for rapid and reliable detection of ICP changes, providing an alternative to invasive intracranial monitoring techniques. Recent studies have demonstrated that ultrasonographic ONSD measurements correlate with direct ICP measurements, making it a valuable tool for clinical decision-making in neurocritical care settings (1-3).
The Glasgow Coma Scale (GCS) is a widely used clinical scoring system for assessing consciousness and neurological function in patients with brain injuries or altered mental status. Several studies have reported a significant association between ONSD and GCS scores in traumatic brain injury, suggesting that patients with lower GCS scores tend to have increased ONSD, which may reflect elevated ICP (4,5). This relationship highlights the potential utility of ONSD measurements as a bedside tool for evaluating neurological deterioration in ICU patients.
Despite the growing body of literature on ONSD and its clinical implications, further research is needed to explore its correlation with various physiological parameters in critically ill patients. In particular, factors such as mechanical ventilation, sepsis, and comorbidities may influence ONSD measurements, warranting a more comprehensive analysis of its diagnostic value (3).
This study aims to investigate the relationship between ONSD and GCS scores in ICU patients while also assessing the impact of key clinical factors, including mechanical ventilation settings, hemodynamic parameters, and comorbidities. By examining these associations, we aim to determine the potential role of ONSD as a non-invasive biomarker for neurological monitoring and patient prognosis in intensive care settings.
Methodology
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and ethical approval was obtained from the institutional ethics committee (IRB Number: 2020.12.208). Informed consent was obtained from all patients or their legal representatives before participation.
Study design and patient selection
This prospective observational study included adult patients (≥18 years old) who were admitted to the intensive care unit (ICU) for various reasons and received treatment between January 2021 and April 2021.
Inclusion and exclusion criteria
Patients were included if they were ≥18 years old and had a hospital stay of at least 24 hours. The exclusion criteria were:
- Age <18 or >75 years,
- Hospital stay <24 hours,
- Presence of conditions that could increase intracranial pressure, such as intracranial mass lesions or intracranial hemorrhage, (Patients with acute intracranial hemorrhage requiring neurosurgical intervention were excluded. However, patients with stable or chronic intracranial hemorrhage were included if no active ICP elevation was present.)
- History of ocular disease, including orbital tumors, optic neuritis, glaucoma, hyperthyroidism, or any condition affecting optic nerve sheath integrity.
Study hypothesis
The primary hypothesis of this study was that lower GCS scores would be associated with higher ONSD values in critically ill patients.
Collected data and study parameters
The study recorded the following parameters for each patient:
- Demographic characteristics (age, gender),
- Body mass index (BMI, kg/m²),
- ICU admission indication,
- Comorbid conditions,
- Vital parameters,
- GCS score,
- Endotracheal intubation status,
- Ventilation mode and ventilation parameters,
- ONSD measurements (mm) at 24 hours and 48-72 hours.
ONSD measurement procedure
ONSD measurements were performed twice for each patient:
- Within the first 24 hours of ICU admission,
- Between 48-72 hours after admission.
Measurements were obtained using ultrasonography by an experienced researcher utilizing a MyLab™Six ultrasound system (Esaote SpA, Genoa, Italy) with a 4–15 MHz linear probe in B-mode. The patient was positioned supine with the head elevated at a 20–30° angle. A gel was applied to the temporal region of the closed eyelid to avoid direct pressure on the eye, and the ultrasound probe was gently placed over the eyelid (Figure 1a).
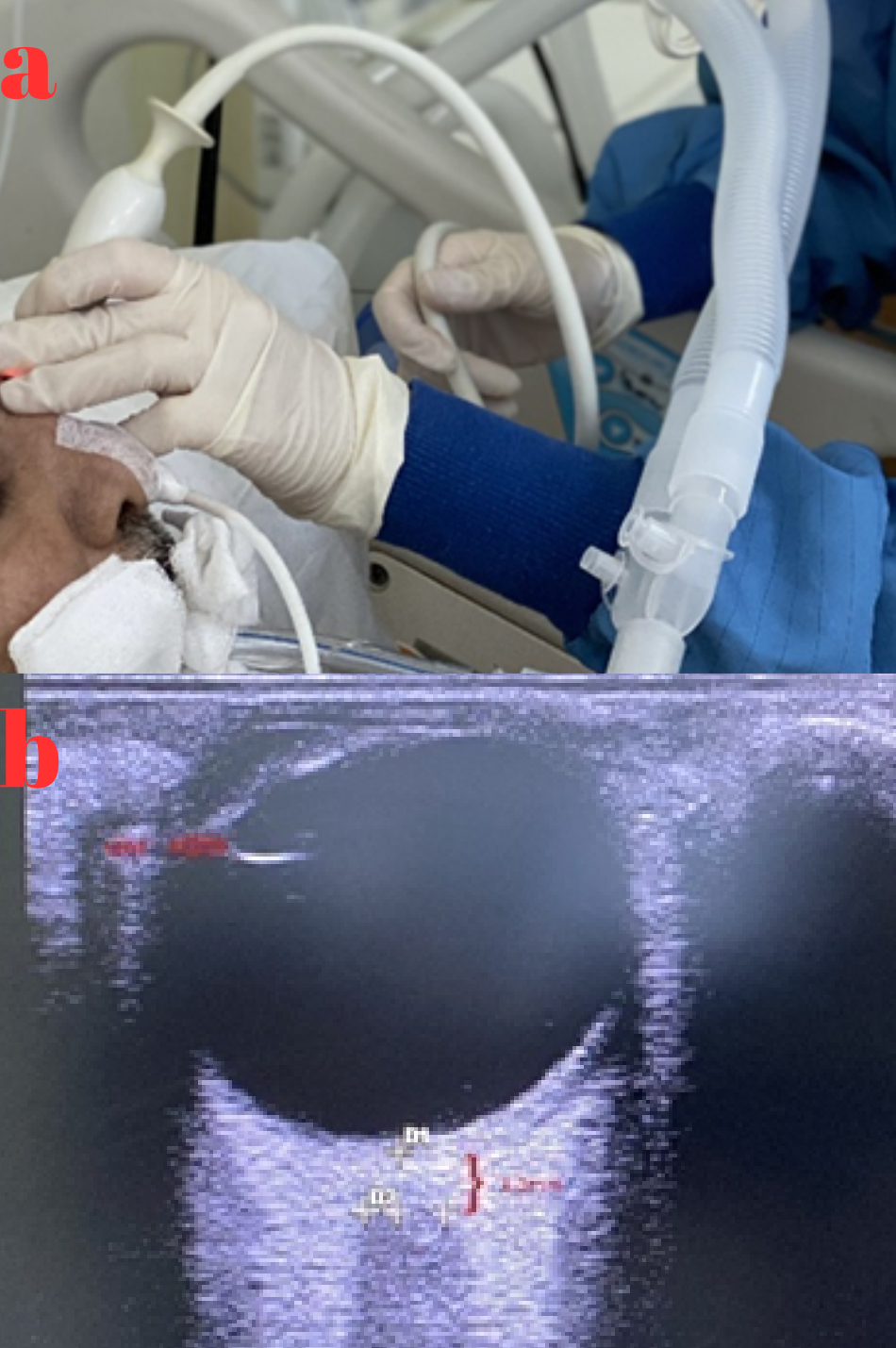
ONSD was measured 3 mm posterior to the retina along the perpendicular axis of the optic nerve using an electronic caliper (Figure 1b). Each measurement was performed twice, and the average value was recorded. All measurements were performed by the same investigator to ensure consistency and reduce interobserver variability.
All intubated patients were ventilated in SIMV mode with a standardized PEEP of 5 cmH2O during ONSD measurements. The No-PEEP group consisted of non-intubated patients who were spontaneously breathing on room air or with supplemental oxygen via face mask without PEEP. All intubated patients received continuous sedation with midazolam (0.02–0.1 mg/kg/h) and remifentanil (0.05–0.2 µg/kg/min) infusions during measurements.
For intubated patients, the verbal component of the Glasgow Coma Scale was recorded as “1T” (for tube), while the eye-opening and motor responses were assessed based on the patient’s reaction to verbal or painful stimuli.
We acknowledge that GCS assessment under sedation provides a suboptimal reflection of neurological status; therefore, to minimize potential bias, patients were analyzed using grouped GCS categories (3–7, 8–11, and 12–15) rather than relying on precise numeric scores. This approach allowed for more consistent interpretation across patients with varying sedation depths and ventilatory condition.
Statistical analysis
The normality of the data was assessed using the Shapiro-Wilk test. We analyzed the relationship between ONSD and gender BMI and Sepsis using independent sample t test. We analyzed the relationship between ONSD and GCS, MAP, Heart Rate, SpO2, PEEP using a linear mixed-effects model with a random intercept for subjects and a compound symmetry covariance structure to account for repeated measurements at 24 and 72 hours. A p-value <0.05 was considered statistically significant.
Sample size calculation
This study aims to compare measurements across three groups based on the GCS levels. A preliminary study with 15 patients was conducted, and the mean values with standard deviations in each group were as follows:
- GCS 3-7 group: 4.1 mm (SD: 0.4)
- GCS 8-11 group: 3.8 mm (SD: 0.4)
- GCS 12-15 group: 3.5 mm (SD: 0.4)
Based on these values, the effect size was calculated as 0.61. To achieve 95% statistical power while maintaining a Type I error rate of 5% (α = 0.05), a power analysis using G*Power determined that at least 45 patients should be included in the study.
However, considering potential patient losses and missing data during follow-up, the study is planned to be conducted with a total of 66 patients to ensure sufficient statistical power and reliability.
Results
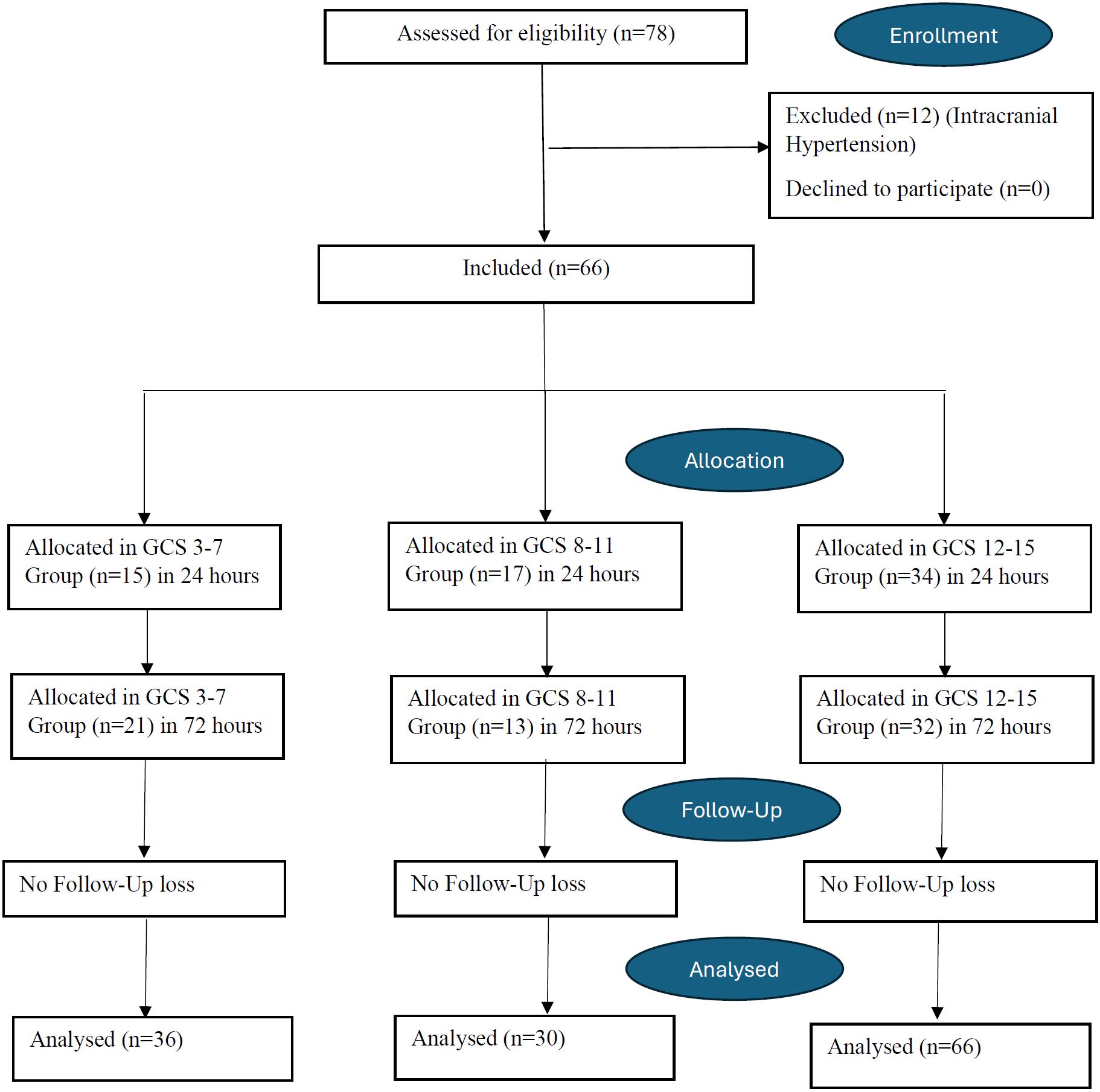
Totally 66 patients were included in this study (Figure 2). In our cohort, the most common indication for intensive care admission was sepsis (n=19), followed by respiratory causes (n=18), including pneumonia, COPD exacerbations, and asthma attacks. Other frequent indications were postoperative close monitoring (n=12), multitrauma (n=8), cerebrovascular events (n=4), and intracranial hemorrhage (n=4). Less frequent causes included status epilepticus (n=1) and intoxication (n=1).
Table 1 presents demographic and clinical characteristics of the study population. Among the 66 patients, 60.6% were male and 39.4% were female. The mean age was 58.67 ± 18.84 years, while the mean BMI was 27.13 ± 5.74. Sepsis was present in 47% of the patients, while 53% had no sepsis.
| Table 1. Demographical data of the patients | ||
|
|
|
|
| Male |
|
|
| Female |
|
|
| Age (years) |
|
|
| BMI (kg/m²) |
|
|
| No Sepsis |
|
|
| Sepsis |
|
|
| Diabetes Mellitus (DM) |
|
|
| Hypertension (HT) |
|
|
| Any Malignancy History |
|
|
| COPD |
|
|
| Chronic Kidney Disease (CKD) |
|
|
| Parkinson’s Disease |
|
|
| Alzheimer’s Disease |
|
|
| Any Cardiac Condition |
|
|
| Patients with No Comorbidities |
|
|
Regarding comorbidities, 24.2% of patients had diabetes mellitus (DM), 31.8% had hypertension (HT), and 12.1% had a history of malignancy. Chronic obstructive pulmonary disease (COPD) was observed in 6.1% of patients, while 13.6% had chronic kidney disease (CKD). Parkinson’s disease and Alzheimer’s disease were present in 3% and 7.6% of patients, respectively. Additionally, 12.1% had a cardiac condition. Notably, 25.8% of the patients had no comorbidities (Table 1).
Table 2 presents the comparison of optic nerve sheath diameter (ONSD) across selected patient characteristics. No statistically significant differences in ONSD were observed between male and female patients (p = 0.655), between patients with BMI < 30 and BMI ≥ 30 (p = 0.191), or between patients with and without sepsis (p = 0.914).
| 1 Independent Sample t Test has been applied. | ||
| Table 2. Comparison of optic nerve sheath diameter between patients characteristics | ||
|
|
|
|
| Male (n=40) |
|
|
| Female (n=26) |
|
|
| BMI<30 (n=51) |
|
|
| BMI>30 (n=15) |
|
|
| No-Sepsis (n=35) |
|
|
| Sepsis (n=31) |
|
|
Table 3 summarizes the results of the linear mixed model analysis examining factors associated with ONSD. PEEP application was strongly associated with higher ONSD values (F = 74.534, p < 0.001). In contrast, mean arterial pressure (MAP), heart rate (HR), and peripheral oxygen saturation (SpO2) categories did not show statistically significant associations with ONSD (all p > 0.05). Timepoint of measurement (24h vs 72h) also had no significant effect on ONSD (F = 0.52, p = 0.472). Importantly, lower Glasgow Coma Scale (GCS) scores were significantly associated with higher ONSD values (F = 54.4, p < 0.001), supporting the primary study hypothesis.
| 1 Linear Mixed Model has been applied. | ||
| Table 3. Comparison of optic nerve sheath diameter between variables | ||
| Fixed Effect | F (df) | p-value1 |
| GCS (per 1-point change) | 54.4 | <0.001 |
| Timepoint (24h vs 72h) | 0.52 | 0.472 |
| HR (beats/min) (<100 vs ≥100) | 3.560 | 0.061 |
| MAP (mmHg) (<65 vs ≥65) | 0.929 | 0.337 |
| SpO₂ (%) (≤97 vs >97) | 1.092 | 0.298 |
| PEEP (cmH₂O) (No vs Yes) | 74.534 | <0.001 |
Discussion
This study aimed to explore the relationship between ONSD and GCS scores in intensive care unit (ICU) patients, while also evaluating the impact of positive end-expiratory pressure (PEEP) on ONSD measurements. Our study demonstrated a significant association between ONSD and Glasgow Coma Scale GCS scores, with lower GCS scores corresponding to higher ONSD values, suggesting a potential role of ONSD as a non-invasive marker of intracranial pressure (ICP) changes in critically ill patients. Patients in the GCS 3–7 group had significantly higher ONSD measurements compared to the GCS 8–11 and GCS 12–15 groups, while the GCS 8–11 group also had significantly higher ONSD values than the GCS 12–15 group (all p < 0.01). Additionally, PEEP application was associated with significantly higher ONSD values compared to patients without PEEP (p < 0.001), supporting previous findings that mechanical ventilation settings may influence ICP dynamics by reducing cerebral venous outflow. In contrast, no significant associations were found between ONSD and MAP, heart rate (HR), SpO2, gender, BMI, or sepsis status, suggesting that these factors do not substantially affect ONSD measurements in ICU patients.
Multiple studies have demonstrated a significant correlation between ONSD and intracranial pressure (ICP), supporting the use of ONSD as a reliable, noninvasive surrogate marker for ICP elevation (7-9). Research has consistently shown that increased ICP leads to ONSD distension due to the continuity of the subarachnoid space around the optic nerve (10,11).
A study by Kshirsagar et al. (2024) demonstrated a significant association between lower GCS scores and increased ONSD in traumatic brain injury (TBI) patients (12). Their findings showed that patients with severe TBI (GCS 2–8) had significantly higher mean ONSD values (6.4 ± 1.0 mm) compared to those with moderate TBI (GCS 9–12, 4.7 ± 0.4 mm), reinforcing the role of ONSD as a potential surrogate marker for elevated intracranial pressure (ICP). Similar to their findings, our study also found a significant correlation between GCS and ONSD, with lower GCS scores being associated with increased ONSD values. These results support the growing body of evidence that ONSD measurement via bedside ultrasonography may serve as a valuable, non-invasive tool for assessing ICP, particularly in critically ill and mechanically ventilated patients. However, while their study included only TBI patients, our study expands this association to a broader ICU population, suggesting that ONSD may be a useful indicator of neurological status beyond the trauma setting.
A study by Wang et al. (2019) demonstrated a strong correlation between ONSD and intracranial pressure (ICP) in traumatic brain injury (TBI) patients undergoing decompressive craniotomy (DC). The study found a significant linear relationship between ONSD and ICP values (r = 0.771, p < 0.0001), with ONSD progressively increasing as ICP rose. The authors identified ONSD cutoff values of 5.48 mm for detecting ICP >13 mmHg and 5.83 mm for ICP >22 mmHg, with high sensitivity and specificity. These findings highlight the utility of ONSD measurement as a noninvasive surrogate for ICP monitoring, offering a reliable alternative to invasive ICP monitoring methods. In line with these findings, our study also observed a significant association between ONSD and markers of neurological status, reinforcing the potential of bedside ultrasonographic ONSD measurement as an adjunctive tool for ICU patients at risk of intracranial hypertension, even in non-TBI populations (13).
A randomized trial by Yanatma et al. (2021) found that PEEP application at 10 cmH2O did not significantly alter ONSD in laparoscopic cholecystectomy patients (14), contrasting with our findings where PEEP was associated with increased ONSD. This discrepancy may be due to differences in patient populations, surgical settings, and baseline intracranial compliance, as their study included healthy ASA I-II patients, whereas our cohort consisted of critically ill ICU patients who may have been more vulnerable to PEEP-induced changes in intracranial pressure. These findings highlight the need for further research in different clinical contexts to clarify the effects of PEEP on ICP.
A study by Fenerci et al. (2024) found that increasing PEEP levels led to a significant rise in ONSD, suggesting an increase in intracranial pressure (ICP), particularly at 10 cmH2O PEEP in patients with midline shift, while PEEP up to 5 cmH2O had no effect (15). Similarly, Balakrishnan et al. (2020) demonstrated that increasing PEEP from 5 to 10 cmH2O and reducing ETCO2 significantly altered ONSD and regional cerebral oxygen saturation (rScO2) in patients with acute traumatic brain injury (TBI), with the highest ONSD observed at 10 cmH2O PEEP and ETCO2 at 40 mmHg (16). While both studies highlight the impact of ventilatory settings on ONSD, our findings suggest that even moderate PEEP levels (not exceeding 8 cmH2O) were associated with increased ONSD in critically ill ICU patients without TBI or midline shift, indicating that ICP changes may not be limited to high PEEP levels or specific neurological conditions. These results emphasize the importance of individualized PEEP management in ICU patients at risk of intracranial hypertension and the need for further research to determine optimal ventilatory strategies across different patient populations.
The generalizability of our findings depends on several factors. First, our study suggests that even moderate levels of PEEP (≤8 cmH2O) can influence ONSD, indicating potential intracranial pressure (ICP) changes in critically ill patients. This finding may be applicable to ICU patients receiving mechanical ventilation, particularly those with impaired cerebral autoregulation. However, our results should be cautiously interpreted in patients with pre-existing neurological conditions, such as traumatic brain injury or intracranial pathology, as their baseline ICP dynamics may differ. Additionally, our study’s methodology, including standardized ultrasound-based ONSD measurements performed by a single investigator, supports reproducibility in similar ICU settings with trained personnel. Given that no significant associations were found between ONSD and other hemodynamic parameters such as MAP, HR, and SpO2, our findings suggest that PEEP-related ONSD changes may be independent of systemic hemodynamics, making them more likely to be generalizable across mechanically ventilated ICU populations. Nonetheless, external validation in larger, multi-center studies with direct ICP measurements is necessary to confirm the broader applicability of our findings across different patient groups and ventilatory conditions.
Our study has several limitations that should be acknowledged. First, the relatively small sample size may limit the generalizability of our findings, and larger studies are needed to confirm our results in a broader population of critically ill patients. Second, the observational nature of our study prevents us from establishing a causal relationship between PEEP application and changes in ONSD, as unmeasured confounding factors may have influenced our results. Third, ONSD measurements were not validated with direct intracranial pressure (ICP) monitoring, such as invasive intraparenchymal or intraventricular pressure measurements, which are considered the gold standard for ICP assessment. While ultrasonographic ONSD measurement is a well-established, non-invasive surrogate for ICP estimation, the lack of direct ICP validation may limit the accuracy of our findings. Fourth, we did not assess the potential impact of other ventilatory parameters, such as driving pressure or tidal volume, which may also influence cerebral hemodynamics and ONSD. Lastly, our study focused on ICU patients, and the applicability of our results to other clinical settings, such as the operating room or emergency department, remains uncertain. Future research should aim to address these limitations by conducting larger, multicenter, randomized controlled trials incorporating direct ICP monitoring and a comprehensive analysis of ventilatory and hemodynamic factors to further elucidate the relationship between PEEP, ONSD, and ICP dynamics.
Conclusion
In conclusion, our findings suggest that increased ONSD is associated with lower GCS scores in ICU patients, indicating a potential role for ONSD measurement in non-invasive ICP assessment. The influence of PEEP on ONSD warrants cautious interpretation, and clinicians should consider individual patient factors when managing ventilatory settings to mitigate potential impacts on intracranial dynamics.
Ethical approval
This study has been approved by the İstanbul S.B.U. Kanuni Sultan Süleyman Training and Research Hospital Clinical Research Ethics Committee (approval date: 10.12.2020, number: 2020.12.208). Written informed consent was obtained from the participants.
Author contribution
Study conception and design: MİG, EİT, EAİ, Oİ, EK, ASŞ; data collection: MİG, EİT, EAİ, Oİ; analysis and interpretation of results: MİG, EİT, EAİ, Oİ; draft manuscript preparation: MİG, EİT, EAİ, Oİ, EK, ASŞ. The author(s) reviewed the results and approved the final version of the article.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Netteland DF, Aarhus M, Smistad E, et al. Noninvasive intracranial pressure assessment by optic nerve sheath diameter: automated measurements as an alternative to clinician-performed measurements. Front Neurol. 2023;14:1064492. https://doi.org/10.3389/fneur.2023.1064492
- Hirzallah MI, Lochner P, Hafeez MU, et al. Optic nerve sheath diameter point-of-care ultrasonography quality criteria checklist: an international consensus statement on optic nerve sheath diameter imaging and measurement. Crit Care Med. 2024;52:1543-56. https://doi.org/10.1097/CCM.0000000000006345
- Lochner P, Czosnyka M, Naldi A, et al. Optic nerve sheath diameter: present and future perspectives for neurologists and critical care physicians. Neurol Sci. 2019;40:2447-57. https://doi.org/10.1007/s10072-019-04015-x
- Zhang X, Ma D, Li W, et al. Correlation between optic nerve sheath diameter measured by bedside ultrasound and intracranial pressure in neurologically ill patients in a Chinese population. BMC Neurol. 2024;24:452. https://doi.org/10.1186/s12883-024-03961-0
- Sekhon MS, McBeth P, Zou J, et al. Association between optic nerve sheath diameter and mortality in patients with severe traumatic brain injury. Neurocrit Care. 2014;21:245-52. https://doi.org/10.1007/s12028-014-0003-y
- Magoon R, Jose J, Suresh V. Research trends in optic nerve sheath diameter monitoring - a bibliometric study. Indian J Anaesth. 2024;68:579-82. https://doi.org/10.4103/ija.ija_205_24
- Robba C, Santori G, Czosnyka M, et al. Optic nerve sheath diameter measured sonographically as non-invasive estimator of intracranial pressure: a systematic review and meta-analysis. Intensive Care Med. 2018;44:1284-94. https://doi.org/10.1007/s00134-018-5305-7
- Kerscher SR, Schöni D, Neunhoeffer F, et al. The relation of optic nerve sheath diameter (ONSD) and intracranial pressure (ICP) in pediatric neurosurgery practice - part II: influence of wakefulness, method of ICP measurement, intra-individual ONSD-ICP correlation and changes after therapy. Childs Nerv Syst. 2020;36:107-15. https://doi.org/10.1007/s00381-019-04336-4
- Fernando SM, Tran A, Cheng W, et al. Diagnosis of elevated intracranial pressure in critically ill adults: systematic review and meta-analysis. BMJ. 2019;366:l4225. https://doi.org/10.1136/bmj.l4225
- Sekhon MS, Griesdale DE, Robba C, et al. Optic nerve sheath diameter on computed tomography is correlated with simultaneously measured intracranial pressure in patients with severe traumatic brain injury. Intensive Care Med. 2014;40:1267-74. https://doi.org/10.1007/s00134-014-3392-7
- Martínez-Palacios K, Vásquez-García S, Fariyike OA, Robba C, Rubiano AM; Noninvasive ICP Monitoring International Consensus Group. Using optic nerve sheath diameter for intracranial pressure (ICP) monitoring in traumatic brain injury: a scoping review. Neurocrit Care. 2024;40:1193-212. https://doi.org/10.1007/s12028-023-01884-1
- Kshirsagar SJ, Pande AH, Naik SV, et al. Bedside ultrasonographic evaluation of optic nerve sheath diameter for monitoring of intracranial pressure in traumatic brain injury patients: a cross sectional study in level II trauma care center in India. Acute Crit Care. 2024;39:155-61. https://doi.org/10.4266/acc.2023.01172
- Wang J, Li K, Li H, et al. Ultrasonographic optic nerve sheath diameter correlation with ICP and accuracy as a tool for noninvasive surrogate ICP measurement in patients with decompressive craniotomy. J Neurosurg. 2019;133:514-20. https://doi.org/10.3171/2019.4.JNS183297
- Yanatma S, Polat R, Sayın MM, Karabayırlı S. The effects of positive end-expiratory pressure (PEEP) application on optic nerve sheath diameter in patients undergoing laparoscopic cholecystectomy: a randomized trial. Braz J Anesthesiol. 2023;73:769-74. https://doi.org/10.1016/j.bjane.2021.12.006
- Fenerci A, Akcil EF, Tunali Y, Dilmen OK. Effect of different positive end expiratory pressure levels on optic nerve sheath diameter in patients with or without midline shift who are undergoing supratentorial craniotomy. Acta Neurochir (Wien). 2024;166:177. https://doi.org/10.1007/s00701-024-06067-1
- Balakrishnan S, Naik S, Chakrabarti D, Konar S, Sriganesh K. Effect of respiratory physiological changes on optic nerve sheath diameter and cerebral oxygen saturation in patients with acute traumatic brain injury. J Neurosurg Anesthesiol. 2022;34:e52-6. https://doi.org/10.1097/ANA.0000000000000706
Copyright and license
Copyright © 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.