Abstract
Paroxysmal sympathetic hyperactivity syndrome (PSH) is a rarely diagnosed syndrome associated with acute brain injury which involves traumatic brain injury, stroke, infectious diseases, and encephalopathy, and results in cyclic episodes of sympathetic and motor hyperactivity (hypertension, tachypnea, hyperthermia, diaphoresis, dystonic posturing). Even though there are some theories regarding the pathophysiology of PSH, the main mechanism still remains unclear. However, it is mostly agreed upon that PSH is caused by an unbalanced activation of the autonomic nervous system caused by the loss of inhibition of excitation in the sympathetic nervous system. Although PSH has been well known for many decades there were misunderstandings regarding the nomenclature and the definition of this syndrome. The term “Paroxysmal Sympathetic Hyperactivity and a diagnostic tool consisted of two different components and named PSH-Assessment Measure (PSH-AM),” was developed to help guiding clinical management in 2014. These components are the diagnostic likelihood tool and the clinical features scale. To diagnose this syndrome first other neurological issues should be ruled out. Once diagnosed in order to treat and avoid secondary brain damage and other adverse outcomes of this syndrome an approach of both pharmacological (such as opioids, B-blockers, alfa2 agonists, benzodiazepines, gabapentin and muscle relaxants) and nonpharmacological should be combined. Pharmacological approach focuses on symptom abortion, prevention and refractory treatment.
Keywords: paroxysmal sympathetic hyperactivity syndrome, traumatic brain injury, acute brain injury
Introduction
Paroxysmal sympathetic hyperactivity (PSH) is a syndrome first identified in 1954 and characterized by episodes of sympathetic discharges, after an acute brain injury either traumatic or non-traumatic (1-4). During this sympathetic discharge episodes; tachycardia, transient fever (hyperthermia), arterial hypertension, tachypnea, diaphoresis, dystonia and posturing (extensor or flexor) may occur spontaneously or after a non-painful stimulus (such as endo-tracheal tube suctioning or passive body positioning) (1,3). PSH is frequently seen in critically ill patients on intensive care units (ICU), with acquired brain injury including traumatic brain injury (80%), hypoxic- ischemic encephalopathy (10%), autoimmune encephalitis associated with N-methyl-D-aspartate receptor antibodies, stroke (mainly hemorrhagic), tumors, cerebrovascular accidents, cerebral fat embolism and infectious diseases such as tuberculous meningitis and usually observed when the patient is started neurological rehabilitation (1-3,5-8).
PSH has been well known for many decades and recognizing this syndrome is very important due to its clinical burden such as longer stay in the ICU, infections, increased morbidity and lengthening of the rehabilitation process (1,9). Even though PSH is clinically very important; there were misunderstandings regarding the nomenclature, definition and the diagnostic criteria of this syndrome (1,9,10). Therefore, a committee came together in 2014 in order to develop a definition and set the diagnostic criteria for the syndrome. So the term “Paroxysmal Sympathetic Hyperactivity” was developed to identify the clinical state seen after severe acquired brain injury of any cause, and resulted in simultaneous paroxysmal transient increases in sympathetic and motor activity (1,3,9,10).
Epidemiology and Risk Factors of PSH
The worldwide yearly incidence of traumatic brain injury (TBI) is 69 million cases (11). While the prevalence of PSH after non-traumatic brain injuries is only 6%; the prevalence of PSH is shown to be between 7,7%- 33% of patients after severe traumatic brain injury, although the real incidence is thought to be much higher (2,4-6,10). Some of the risk factors for PSH include; male gender, young age, early onset of hyperthermia, GCS score, tracheostomy, the degree of diffuse axonal injury, mid-brain and pontine lesions, the cause and the severity of the brain injury (mostly after traumatic brain injury(79,4%), hypoxic encephalopathy and stroke (3-5,12-14).
Pathophysiology of PSH
Basically PSH is thought to be caused by the imbalance of the autonomic nervous system (ANS) (4). After damage to the brain ANS activation leads to immediate inflammation and metabolic response. This response results in the core sympathetic symptoms of PSH if the parasympathetic mechanisms cannot compensate (6). However, the main pathophysiologic mechanisms behind PSH, even though there are some theories, still remains unclear. These theories are the disconnection theory, excitatory/inhibitory ratio (EIR) model, the neuroendocrine dysregulation theory and the neutrophil extracellular traps theory (2,12).
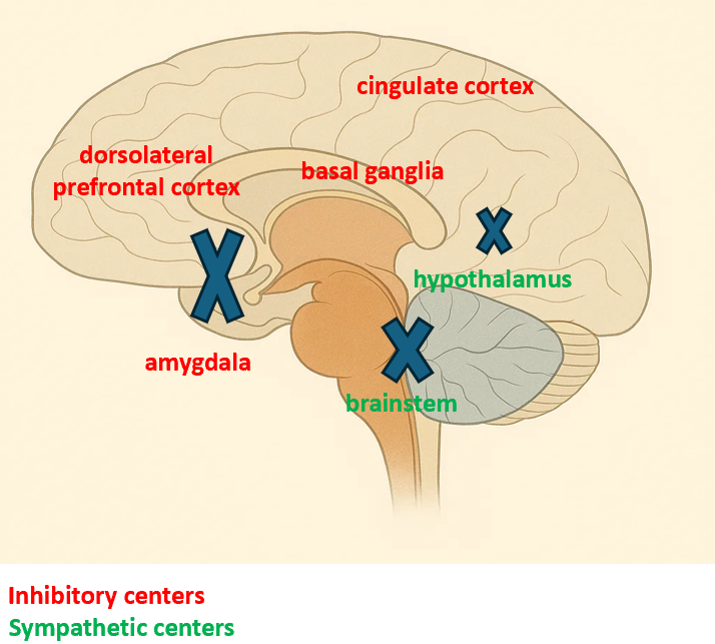
Disconnection theory (Figure 1) supports a disconnection related to focal or diffuse brain damage, either anatomical or functional, between cortical inhibitory centers; which are the hippocampus, insular cortex, cingulate cortex, middle temporal cortex, amygdala, basal ganglia and the dorsolateral prefrontal cortex and the sympathetic centers which are; the hypothalamus, brain stem and mesencephalon (2,3,6,10,12). When there is a disconnection, losing the cortical inhibition results in an inflated sympathetic response to normally non-painful stimuli. However the problem with this model is that it does not explain the episodic nature of this syndrome (2).
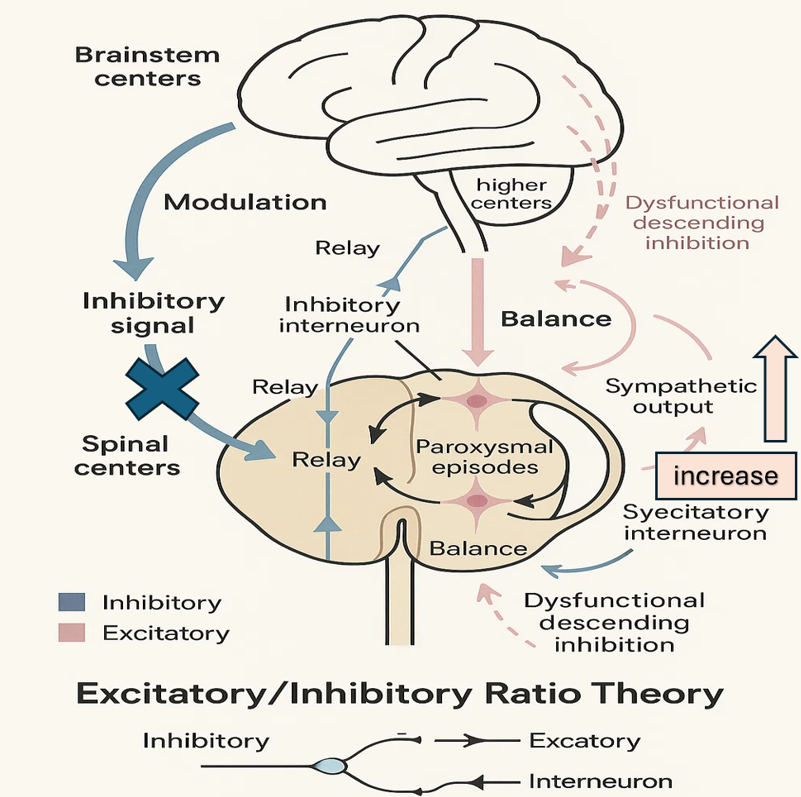
Excitatory/inhibitory ratio model (Figure 2) helps explain the hypersensitivity of the reactions and the episodic nature of PSH (5,12). This theory, being the mostly agreed upon, supports an unbalanced relationship between inhibitory (brainstem and diencephalon) and the excitatory (spinal cord) pathways. In first place we witness the loss of descending inhibition which leads to excitation of the spinal circuits against non-painful stimuli resulting in hyperactive sympathetic responses (2,6,10,12). Other mechanisms try to compensate for the loss of inhibitory control, but they contain partial recovery of the descending pathways, and an increase in gamma-aminobutyric acid levels. Because these mechanisms are episodic in nature we can explain the paroxysmal characteristic of PSH (2).
Neuroendocrine dysregulation theory refers to a dysfunction in the hypothalamic–pituitary–adrenal (HPA) axis. PSH causes overactivation of HPA axis resulting in an uncontrolled adrenergic outflow which in turn leads to an increased amount of catecholamines in the circulation (2,3). During the sympathetic discharge episodes blood levels of epinephrine, norepinephrine, dopamine and adrenocorticotropic hormone (nearly 40%) rises significantly, favoring the involvement of neuroendocrine system in the pathophysiology of PSH (2,12).
Neutrophil extracellular traps (NETs) theory explains the infiltration of paraventricular nucleus with neutrophils after TBI. The release of NETs leads to microglial activation and interleukin 1 B release resulting in sympathetic activation and a change in neurotransmitter levels (15). Cerebral contusion, brain edema, cerebral ischemia may also result in the release of excitatory amino acids at a cellular level intensifying the development of PSH (12).
Diagnosing PSH
In order to diagnose PSH, like all other syndromes, first one must have a strong degree of suspicion. First step of diagnosing PSH is excluding all the other causes of the symptoms such as sepsis, pulmonary embolism, epilepsy or hypoxic brain (1,2,6,10). As mentioned above these findings of sympathetic hyperactivity episodes are tachycardia, hypertension, hyperthermia, sweating, tachypnea, posturing (2,5,6,9,10). Moreover some of the less frequent symptoms are mydriatic pupils, altered consciousness, bowel disorders, alterations in blood glucose levels, increased bronchial secretions and hypersalivation, desynchrony with mechanical ventilator, excitation and horripilation (2,5,6). The duration of these episodes may vary between minutes, hours, and in severe cases it may occur a few times in a day. They may even become continuous in refractory cases (4). Because these symptoms may overlap with other TBI related pathological outcomes such as; seizures, sepsis, embolism, neuroleptic malignant syndrome, central fever, traumatic pain, different brain injuries, sedation withdrawal, hypoxia, rhabdomyolysis, and dehydration diagnosing PSH is often delayed or misdiagnosed resulting in complications in the management of the case (1,3,4). There is also another entity resembling paroxysmal sympathetic hyperactivity in nomenclature and that is autonomic dysreflexia. Autonomic dysreflexia patients have episodes of hypertension after a high spinal cord (above the level of thoracal 6 vertebra) injury, however they are usually bradycardic as a reflex response to hypertension, and they often have debilitating headaches; differentiating this syndrome from PSH (16-20).
Diagnosing PSH as early as possible has great importance because delayed recognition may result in unnecessary work-up and inappropriate use of medications may prolong hospitalization. Uncontrolled symptoms can lead to secondary brain injury caused by hypertension, hyperthermia and cardiac damage. It should also be kept in mind that these other pathologies may coexist and even trigger episodes of PSH (4).
Therefore the intensivist should keep the possibility of PSH in mind, especially when the patient is coming out of a coma or when the patient is weaned from sedatives (4,10).
Neuro-imaging techniques like computerized tomography (for acute phase) and magnetic resonance images (in stable patients) are helpful to identify the anatomical place and the morphological structure of the injury and as in a case report from the literature, they often show diffuse axonal injury (3,6,10,12,21). Aside from these techniques, dysconnectivity in white matter; especially on the right posterior internal capsule, corpus callosum and the splenium shown on diffusion tensor imagings are associated with the early stages of PSH (2).
Even though there is no specific biomarker for PSH a study by Fernandez et al showed that plasma catecholamine (and to a lesser extend adrenocortical hormone) levels increased during the episodes of PSH (22). Moreover another study showed that procalcitonin levels can be used to differentiate between infectious pathologies and PSH (23).
A diagnostic tool named Paroxysmal Sympathetic Hyperactivity - Assessment Measure which consisted of two different components, was developed in order to help guiding the clinical management and to standardize the diagnostic criteria. One of the components, referring to the possibility of the diagnosis, is a diagnosis likelihood tool (DLT) (Table 1) derived from 11 characteristics of this syndrome, and the other one, evaluating the severity and intensity of the symptoms in each hyperactivity episode, is a clinical feature scale (CFS) (Table 2) (1,3,9). The DLT evaluates 11 main items with the presence of each scored as one point and absences are scored as zero. If the symptoms persist for three consecutive days, the diagnosis of PSH is more probable. Data to evaluate six main symptoms CFS is used. CFS has a value range between zero and three. A combined score of DLT and CFS gives us the Paroxysmal Sympathetic Hyperactivity Assessment Measure (PSH-AM) (Table 3). If the score is < 8 points the diagnosis is unlikely, if it is between 8-16 points the diagnosis is possible and if the score is > 17 points the diagnosis is probable (1,2,9).
| Table 1. Diagnostic likelihood tool (1 point for each feature) | |
| Clinical features occur simultaneously | 1 point |
| Episodes are paroxysmal in nature | 1 point |
| Sympathetic over – reactivity to normally non-painful stimuli | 1 point |
| Features persist > 3 consecutive days | 1 point |
| Features persist > 2 weeks post brain injury | 1 point |
| Features persist despite treatment of alternative differential diagnosis | 1 point |
| Medication administered to decrease sympathetic features | 1 point |
| > 2 episodes daily | 1 point |
| Absence of parasympathetic features during episodes | 1 point |
| Absence of other presumed cause of features | 1 point |
| Antecedent acquired brain injury | 1 point |
| Table 2. Clinical feature scale | ||||
|
|
|
|
|
|
| Heart Rate |
|
|
|
|
| Respiratory Rate |
|
|
|
|
| Systolic Blood Pressure |
|
|
|
|
| Temperature |
|
|
|
|
| Sweating |
|
|
|
|
| Posturing During Episodes |
|
|
|
|
| Table 3. Paroxysmal sympathetic hyperactivity – assessment measure (PSH-AM) | |
| Unlikely | <8 points |
| Possible | 8-16 points |
| Probable | >17 points |
Treatment Modalities in PSH
Treating PSH is challenging due to the lack of guidelines and the complicated etiology of the syndrome. That is why the treatment modalities mostly focus on controlling the symptoms and the episodes (2,4).
Treatment approach should include adequate sedation, analgesia and muscle relaxation. However, the clinician must be ready to treatment related scenarios like prolonged mechanical ventilation support and a delay in neurological rehabilitation of the patient (3). Moreover, while treating PSH the possibility of multisystem organ dysfunction must also be considered and possible adverse events like cardiac hypertrophy, muscle wasting and dehydration must also be treated. We must also make sure that the patient is receiving enough calories and water- electrolyte levels are balanced (6,10). In order to treat this syndrome, an approach of both pharmacological and nonpharmacological should be combined (4).
Pharmacological Treatment
Medical treatments include a diversity of drug groups, each affecting or preventing different symptoms. Opioids, beta-blockers, dopamine agonists, alfa2 agonists (clonidine, dexmedetomidine), GABAergic agents, benzodiazepines, gabapentin, baclofen (can be given either enterally or intrathecal) and muscle relaxants are mostly used (1,3,4,10). Pharmacological approach focuses on three steps: symptom avoidance, prevention and refractory treatment. Symptom abortive medications should be administered as soon as the episodes are identified they focus on stopping the paroxysmal episodes (2). They have a rapid onset of action and a short half-life; for example, use of antipyretics to treat hyperthermia, sedatives for agitation, muscle relaxants to relieve spasticity and postures, and antihypertensive agents for hypertension. Morphine and short acting benzodiazepines are considered first line treatment (2,4).
Preventive medications aim to control the episodes and to reduce their frequency as well as severity, in order to avoid any arrhythmia, cardiac overload, dehydration, and loss of muscle mass. This approach includes the usage of non-selective beta-blockers, alfa 2 agonists bromocriptine, baclofen, gabapentin and long-acting benzodiazepines. Among these drugs, Propranolol a non-selective B-blocker has shown to reduce mortality rates when given 20-60mg doses in every 4-6 hours. Besides its cardiovascular effects, propranolol also helps lower the body temperature. Furthermore, dexmedetomidine, an alpha-2 agonist, can be administered as an intravenous infusion at a rate of 0.2–0.7 mcg/kg/h to provide sedation and analgesia, regardless of whether the patient is receiving ventilatory support or not. It is also preferred on TBI patients to prevent PSH (2,4).
Refractory treatment modalities aim to avoid secondary brain damage from hypertension, hyperthermia and cardiac damage. During the refractory period continues or bolus intravenous medications likes propofol, benzodiazepines, opioids or dexmedetomidine can be used. Also, intrathecal baclofen use may also be considered in some refractory cases in which posturing episodes persist. Dantrolene, a ryanodine receptor antagonist may also be used for posturing during the refractory period, but with close monitorization of liver function tests (3,4,6).
Because there is no treatment guideline, centers can prepare their own PSH treatment protocols according to clinical presentations of PSH (3,4). Besides, we can also use the PSH-AM scores to combine treatment modalities. For patients with a score of <8 points only the dominant symptoms might be targeted, whereas for patients with a score of 8-16 points both symptomatic and preventive modalities might be used. Patients with a score of >17 points might need symptomatic, preventive treatment as well as baclofen, gabapentin and even dantrolene (6).
Non-pharmacological treatment
As mentioned above, the treatment of PSH should include both pharmacological and non-pharmacological approaches. Non-pharmacological approach aims to prevent any sympathetic hyperactivity episode triggering events, reduce sympathetic nervous activity and lighten any adverse effects (3).
Avoidance of excessive external stimuli while giving daily care, and controlling room temperature are of major importance (3,5).
During ICU management of patients with PSH it must be taken into account that energy consumption of these patients are increased during the episodes, therefore their caloric requirements are also increased because of a hyperfunctioning brain (24,25). Close monitorization of nutrition and hydration status as well as early initiation of enteral nutrition is of crucial importance (26).
Physiotherapy must also keep in mind in order to extend the motion range and prevent contractile postures (3).
Family support and their education are also very important for the management of these patients because the road to recovery is hard and long. Also, Salmani et al showed that family related stimulations are more effective than sensory stimulations in order to improve consciousness (24,27).
The relationship of PSH with cerebral hypoxia might bring hyperbaric oxygen therapy (HBOT) to mind. It was shown in a case series HBOT controlled autonomic discharges and posturing episodes in the subacute phase of TBI and is a good treatment option in refractory PSH with pharmacological therapy (28). Moreover, in a study conducted by Wang et al, two groups of PSH patients, one received HBOT, and the other group did not, were compared. The study revealed that the group which had HBOT had lower PSH-AM scores, more clear relief of PSH symptoms, more awake patients, higher Glasgow Coma Scale scores and shorter ICU stay (29).
Clinical Features and Intensive Care Management of PSH
Paroxysmal Sympathetic Hyperactivity syndrome is a complex disease in which the main symptoms consist of paroxysmal episodes of sympathetic and motor hyperactivity. Although these main symptoms are tachycardia, tachypnea, hypertension, hyperthermia, sweating and posturing, it should also be expected to see different symptoms in each patient (3). During the ICU stay these hyperactivity episodes occur in a cyclic pattern. Even though the episodes may occur spontaneously, they may also occur after non-painful stimuli such as endotracheal tube aspirating, exposure to touch or light or physiotherapy (6).
Another important approach is to remember that some of the symptoms might be masked by the therapeutic processes of TBI or may overlap with other neurological issues related to TBI (3,4). Number of symptoms rather than the duration of episodes are more indicative as to the severity of the disease. Non-adequate controlling of these symptoms may result in secondary brain damage and therefore leading to a poor prognosis (4,6).
The episodes may last a few minutes or as long as two hours, and they can even occur several times in a day or they may occur continuously in refractory cases (3,4). On the other hand the duration of PSH may also change between a few weeks and more than a year (3).
PSH occurs in three stages. First one, the hyperacute phase usually refers to the first week of admission in which the damage to the brain is maximum but the patient is usually asymptomatic due to heavy sedation, analgesia and neuromuscular blockers (if needed) in order to treat TBI and other related injuries. This results in a delay in diagnosis unless the patient awakens for some reason. The second phase is the one in which the patient shows paroxysmal episodes of sympathetic and motor discharges. The last phase starts when hyperhidrosis ends. During this stage declines in posturing episodes are seen. Furthermore, this stage represents the rehabilitation phase which can last for years but with less frequent and severe episodes (6,10).
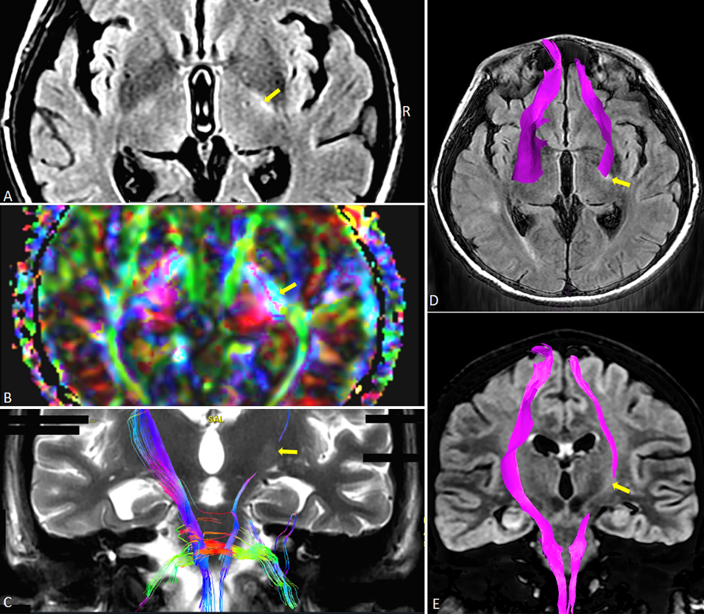
It should also keep in mind that other medical conditions related to trauma and intensive care treatments may also cause delays in diagnosing PSH, as in a case reported by ourselves (21). In this case report it was stated that a 26-year-old female suffering from TBI was admitted to the ICU with diffuse axonal injury and deep coma. Even though she had episodes of sympathetic activities at early admission she also had other medical problems like sepsis and acute hypoxemic respiratory failure at the same time. During her stay in the ICU her magnetic resonance images showed diffuse axonal injury (Figure 3) and the sympathetic symptoms re-occurred while there were no other contemporaneous medical problems. Since PSH-AM was applied. Her CFS was 13, DLT was 9 and she had a total PSH-AM score of 22 and diagnosed with PSH. The diagnostic tool PSH-AM can help the clinician diagnose and treat PSH at early stages, if it begins to be used from the admission of the patient unless there are no other concurrent pathologies.
PSH is often recognized once the intravenous sedation infusions are stopped and the patient begins to awaken. In the case report mentioned above the patient was given metoprolol, gabapentin, tramadol, dexmedetomidine, amantadine sulfate, melatonin and modafinil as pharmacological treatment to control the symptoms (21).
Paroxysmal Sympathetic Hyperactivity syndrome is usually associated with poor long-term outcomes such as, low level of consciousness and low Glasgow Coma Scale scores, longer hospital stays, higher dependance to mechanical ventilation, tracheostomy indication, and more infectious complications (3,5,30). While managing these patients in the ICU it must be kept in mind that PSH results in a hypermetabolic state which leads to weight loss and dehydration as a result of hypercatabolism, heterotopic ossifications, inflammation, immunosuppression and infections, all of which resulting in excessive interventions, delayed rehabilitation, increase in morbidity, longer hospital and ICU stays (1-3,6,30). Even though there is no consensus on the relationship of PSH and weaning from mechanical ventilation, the intensivist should also evaluate the effects of PSH on the weaning process (2).
Conclusion
Prompt recognition of PSH is essential, because delayed diagnosis may result in unnecessary work-up, inappropriate use of medications, excessive interventions, and prolonged hospitalization. The occurrence of PSH after TBI should always be suspected especially if the patient is showing episodes of sympathetic hyperactivity while being weaned from sedative agents. Because PSH is often recognized once the intravenous sedation infusions are stopped and the patient begins to awaken. In order to treat this syndrome an approach of both pharmacological and nonpharmacological should be combined. It must be kept in mind that uncontrolled symptoms may also lead to secondary brain injury caused by hypertension, hyperthermia, cardiac damage and even death.
Author contribution
Study conception and design: TU, AGYS; data collection: AGYS, ZF; analysis and interpretation of results: AGYS, TU; draft manuscript preparation: AGYS, TU. The author(s) reviewed the results and approved the final version of the article.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Godo S, Irino S, Nakagawa A, et al. Diagnosis and management of patients with paroxysmal sympathetic hyperactivity following acute brain injuries using a consensus-based diagnostic tool: a single institutional case series. Tohoku J Exp Med. 2017;243:11-8. https://doi.org/10.1620/tjem.243.11
- Panjaitan JA, Ginting LF, Rajagukguk M, Sibagariang DB, Siahaan AM. Paroxysmal sympathetic hyperactivity: current update on diagnosis, treatments, and outcomes. Surg Neurol Int. 2025;16:163. https://doi.org/10.25259/SNI_1130_2024
- Zheng RZ, Lei ZQ, Yang RZ, Huang GH, Zhang GM. Identification and management of paroxysmal sympathetic hyperactivity after traumatic brain injury. Front Neurol. 2020;11:81. https://doi.org/10.3389/fneur.2020.00081
- Samuel S, Allison TA, Lee K, Choi HA. Pharmacologic management of paroxysmal sympathetic hyperactivity after brain injury. J Neurosci Nurs. 2016;48:82-9. https://doi.org/10.1097/JNN.0000000000000207
- Totikov A, Boltzmann M, Schmidt SB, Rollnik JD. Influence of paroxysmal sympathetic hyperactivity on the functional outcome of neurological early rehabilitation patients: a case control study. BMC Neurol. 2019;19:162. https://doi.org/10.1186/s12883-019-1399-y
- Godoy D, Panhke P, Guerrero Suarez PD, Murillo-Cabezas F. Paroxysmal sympathetic hyperactivity: an entity to keep in mind. Medicina Intensiva (English Edition). 2019;43:35-43. https://doi.org/10.1016/j.medine.2018.10.003
- Wang D, Su S, Tan M, Wu Y, Wang S. Paroxysmal sympathetic hyperactivity in severe Anti-N-Methyl-d-Aspartate receptor encephalitis: a single center retrospective observational study. Front Immunol. 2021;12:665183. https://doi.org/10.3389/fimmu.2021.665183
- Li Z, Chen W, Zhu Y, et al. Risk factors and clinical features of paroxysmal sympathetic hyperactivity after spontaneous intracerebral hemorrhage. Auton Neurosci. 2020;225:102643. https://doi.org/10.1016/j.autneu.2020.102643
- Baguley IJ, Perkes IE, Fernandez-Ortega JF, et al. Paroxysmal sympathetic hyperactivity after acquired brain injury: consensus on conceptual definition, nomenclature, and diagnostic criteria. J Neurotrauma. 2014;31:1515-20. https://doi.org/10.1089/neu.2013.3301
- Meyer KS. Understanding paroxysmal sympathetic hyperactivity after traumatic brain injury. Surg Neurol Int. 2014;5:490-2. https://doi.org/10.4103/2152-7806.144632
- Burzynska M, Wozniak J, Urbanski P, et al. Heart rate variability and cerebral autoregulation in patients with traumatic brain injury with paroxysmal sympathetic hyperactivity syndrome. Neurocrit Care. 2025;42:864-77. https://doi.org/10.1007/s12028-024-02149-1
- Xu SY, Zhang Q, Li CX. Paroxysmal sympathetic hyperactivity after acquired brain injury: an integrative review of diagnostic and management challenges. Neurol Ther. 2024;13:11-20. https://doi.org/10.1007/s40120-023-00561-x
- Qian J, Min X, Wang F, Xu Y, Fang W. Paroxysmal sympathetic hyperactivity in adult patients with brain injury: a systematic review and meta-analysis. World Neurosurg. 2022;166:212-9. https://doi.org/10.1016/j.wneu.2022.03.141
- Perkes I, Baguley IJ, Nott MT, Menon DK. A review of paroxysmal sympathetic hyperactivity after acquired brain injury. Ann Neurol. 2010;68:126-35. https://doi.org/10.1002/ana.22066
- Zhu K, Zhu Y, Hou X, et al. NETs lead to sympathetic hyperactivity after traumatic brain injury through the LL37-Hippo/MST1 pathway. Front Neurosci. 2021;15:621477. https://doi.org/10.3389/fnins.2021.621477
- Jafari AA, Shah M, Mirmoeeni S, et al. Paroxysmal sympathetic hyperactivity during traumatic brain injury. Clin Neurol Neurosurg. 2022;212:107081. https://doi.org/10.1016/j.clineuro.2021.107081
- Morgan S. Management of autonomic dysreflexia in the community. Br J Community Nurs. 2020;25:496-501. https://doi.org/10.12968/bjcn.2020.25.10.496
- Solinsky R, Kirshblum SC, Burns SP. Exploring detailed characteristics of autonomic dysreflexia. J Spinal Cord Med. 2018;41:549-55. https://doi.org/10.1080/10790268.2017.1360434
- Hou S, Rabchevsky AG. Autonomic consequences of spinal cord injury. Compr Physiol. 2014;4:1419-53. https://doi.org/10.1002/cphy.c130045
- Eldahan KC, Rabchevsky AG. Autonomic dysreflexia after spinal cord injury: systemic pathophysiology and methods of management. Auton Neurosci. 2018;209:59-70. https://doi.org/10.1016/j.autneu.2017.05.002
- Gizem A, Selimoğlu Y, Aytaç E, Nizam B, Utku T. Diagnosing paroxsymal sympathetic hyperactivity syndrome after traumatic brain injury, an inspiring case report. Paper presented at: 24th International Intensive Care Symposium; 2023 May 2-3; İstanbul, Türkiye.
- Fernandez-Ortega JF, Baguley IJ, Gates TA, Garcia-Caballero M, Quesada-Garcia JG, Prieto-Palomino MA. Catecholamines and paroxysmal sympathetic hyperactivity after traumatic brain injury. J Neurotrauma. 2017;34:109-14. https://doi.org/10.1089/neu.2015.4364
- Bindra A, Chowdhary V, Dube SK, Goyal K, Mathur P. Utility of serum procalcitonin in diagnosing paroxysmal sympathetic hyperactivity in patients with traumatic brain injury. Indian J Crit Care Med. 2021;25:580-3. https://doi.org/10.5005/jp-journals-10071-23811
- Yin P, Pan Y, Chen D, et al. Diagnosis and management of paroxysmal sympathetic hyperactivity: a narrative review of recent literature. Eur J Med Res. 2025;30:349. https://doi.org/10.1186/s40001-025-02564-w
- Mehta NM, Bechard LJ, Leavitt K, Duggan C. Severe weight loss and hypermetabolic paroxysmal dysautonomia following hypoxic ischemic brain injury: the role of indirect calorimetry in the intensive care unit. JPEN J Parenter Enteral Nutr. 2008;32:281-4. https://doi.org/10.1177/0148607108316196
- Caldwell SB, Smith D, Wilson FC. Impact of paroxysmal sympathetic hyperactivity on nutrition management after brain injury: a case series. Brain Inj. 2014;28:370-3. https://doi.org/10.3109/02699052.2013.865265
- Salmani F, Mohammadi E, Rezvani M, Kazemnezhad A. The effects of family-centered affective stimulation on brain-injured comatose patients’ level of consciousness: a randomized controlled trial. Int J Nurs Stud. 2017;74:44-52. https://doi.org/10.1016/j.ijnurstu.2017.05.014
- Lv LQ, Hou LJ, Yu MK, Ding XH, Qi XQ, Lu YC. Hyperbaric oxygen therapy in the management of paroxysmal sympathetic hyperactivity after severe traumatic brain injury: a report of 6 cases. Arch Phys Med Rehabil. 2011;92:1515-8. https://doi.org/10.1016/j.apmr.2011.01.014
- Wang H, Li Y, Shen S, et al. Hyperbaric oxygen therapy for paroxysmal sympathetic hyperactivity syndrome after brain injury: a multicenter, retrospective cohort study. Med Gas Res. 2025;15:327-31. https://doi.org/10.4103/mgr.MEDGASRES-D-24-00077
- Nasa P, Majeed NA, Juneja D. Paroxysmal sympathetic hyperactivity after traumatic brain injury: current understanding and therapeutic options. Indian J Crit Care Med. 2024;28:97-9. https://doi.org/10.5005/jp-journals-10071-24647
Copyright and license
Copyright © 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.