Abstract
Patients infected with the 2019 novel coronavirus (COVID-19) predominantly present with respiratory symptoms. In severe cases, COVID-19 creates a prothrombotic state that significantly increases the risk of thromboembolic events. These complications are primarily in the form of venous thromboembolism, with a cumulative incidence of up to 49% among hospitalized patients. In contrast, arterial thromboembolic events such as ischemic stroke and myocardial infarction have been rarely reported in the literature. In this case report, we present a case of aortic thrombosis complicated by peripheral embolism and/or thrombosis as an example of arterial thromboembolism.
Keywords: aortic thrombosis, peripheral embolism, COVID-19 associated thrombosis, arterial thromboembolism
Introduction
In December 2019, the novel coronavirus (COVID-19) was discovered and identified in the viral pneumonia cases in Wuhan, Hubei Province, China (1).
Among the several coronaviruses, two are associated with severe clinical symptoms: severe acute respiratory syndrome (SARS) coronavirus (SARS-CoV), and Middle East respiratory syndrome (MERS) coronavirus (MERS-CoV) (2). The clinical manifestation and severity of COVID-19 are similar to those of SARS-CoV. At least 20.5% of cases had one underlying disease, including hypertension, diabetes mellitus, coronary heart disease, chronic obstructive pulmonary disease, cerebrovascular disease, and chronic liver disease. Common indicator abnormalities include decreased white blood cell count (41.0%), increased C-reactive protein (75.2%) and lactate dehydrogenase (LDH) levels (23.6%), and decreased lymphocyte count (26.1%) (3).
Fever was present in 43.8% of the patients on admission, but developed in 88.7% during hospitalization. The second most common symptom was cough (67.8%); nausea or vomiting (5.0%) and diarrhea (3.8%) were uncommon (4).
By October 2024, over 776 million confirmed cases and over 7 million deaths have been reported globally (5).
Up to 14% of infected patients develop interstitial pneumonia, which may progress to acute respiratory distress syndrome needing intensive Care Unit (ICU) care. Evidence indicates severe COVID-19 is linked to a pro-hemostatic state affecting thromboembolism risk. A pooled analysis showed that rising D-dimer levels predict adverse outcomes, indicating coagulopathy, with higher D-dimer in severe cases than milder ones (6).
A study found a higher risk of pulmonary embolism in COVID-19 ARDS patients than other ARDS causes, even with thromboprophylaxis. Venous thromboembolism (VTE) occurrence was about 20%, reaching 49% during hospitalization. A COVID-19 thrombosis guide recommends a baseline CT chest and sequential D-dimer monitoring. Few arterial thromboembolism cases, like stroke or myocardial infarction, were reported (6).
This article presents a case of aortic thrombosis complicated by peripheral embolism and/or thrombosis.
Case Presentation
A 66-year-old male patient with a 5-year history of hypertension presented to our hospital’s COVID-19 outpatient clinic on August 5, 2020, with complaints of fever, cough, and shortness of breath. The patient tested positive for COVID-19 via a PCR test from a nasopharyngeal swab and was admitted to the hospital with a diagnosis of COVID-19 pneumonia for monitoring.
On the second day of hospitalization, due to worsening respiratory difficulty and oxygen saturation (Sat O2) remaining below 90% despite the administration of oxygen via nasal cannula at 10 L/min, the patient was transferred to ICU. Upon ICU admission, the patient received 14 L/min nasal oxygen, and the Glasgow Coma Scale (GCS) was E4M6V5. Vital signs were: respiratory rate 42/min, blood pressure 145/82 mmHg, pulse rate 98/min, and temperature 37.4°C.
Laboratory findings revealed the following:
- Blood glucose: 153 mg/dL (70–110 mg/dL)
- Urea: 16.82 mg/dL (7–20 mg/dL)
- Creatinine: 0.87 mg/dL (0.51–0.95 mg/dL)
- CRP: 198.4 mg/L (0–5 mg/L)
- LDH: 498 U/L (0–247 U/L)
- Procalcitonin: 0.55 ng/mL (0–0.1 ng/mL)
- D-dimer: 910 ng/mL (190–500 ng/mL)
- Ferritin: 1151.6 ng/mL (10–291 ng/mL)
Arterial blood gas analysis revealed:
- pH: 7.518 (7.35–7.45)
- pCO2: 20.7 mmHg (35–45 mmHg)
- pO2: 53.3 mmHg (83–108 mmHg)
- HCO3: 16.7 mmol/L (22–26 mmol/L)
- SpO2: 82.6% (95–99%)
- Lactate: 2 mmol/L (0.5–1.6 mmol/L)
Complete blood count results were:
- WBC: 7.67 × 10^9/L (4.23–9.07 × 10^9/L)
- Hgb: 126 g/L (137–175 g/L)
- Hct: 36.2% (40.1–51%)
- Platelets: 297 × 10^9/L (160–340 × 10^9/L)
- Neutrophils: 6.70 × 10^9/L (1.78–5.38 × 10^9/L)
- Lymphocytes: 0.62 × 10^9/L (1.32–3.57 × 10^9/L)
Coagulation parameters revealed:
- PT: 14.5 s (9–15 s)
- INR: 1.14 (0.8–1.2)
- APTT: 27.8 s (22–38 s)
- Fibrinogen: 645.14 mg/dL (60–150 mg/dL)
The patient was treated with Favipiravir 200 mg 2 × 3 tablets, Gemifloxacin 320 mg 1 × 1 tablet, Dexamethasone 6 mg once daily, Enoxaparin 40 mg twice daily, Pantoprazole 40 mg twice daily, and Isolyte at 20 cc/h. The patient was monitored with 14 L/min nasal oxygen, 4×2 hours of noninvasive ventilation, and intermittent prone positioning. On the 5th day of ICU admission, the patient received the first dose of Tocilizumab 400 mg, followed by a second dose 48 hours later.
On the 8th day, the patient was intubated and connected to mechanical ventilation due to respiratory failure and was monitored with FiO2 at 90%. On the 9th day, antibiotic treatment was adjusted based on the consultation of infectious diseases, including Meropenem 1 g 3×1, Linezolid 600 mg 2×1, and Tigecycline 50 mg 2×1. Due to reduced urine output, hemodialysis was initiated on the 10th day, while enteral feeding was maintained. Subsequently, urine output improved.
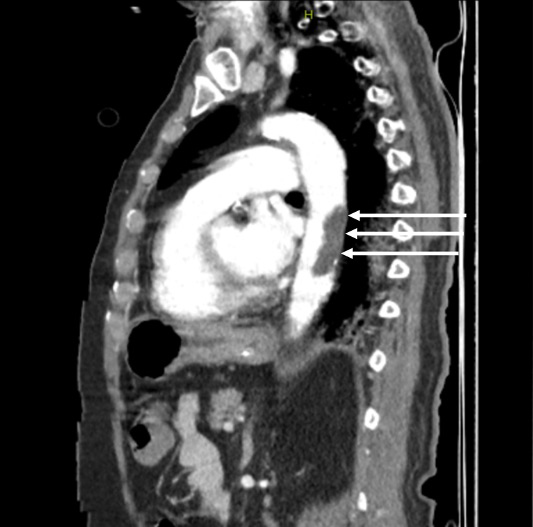
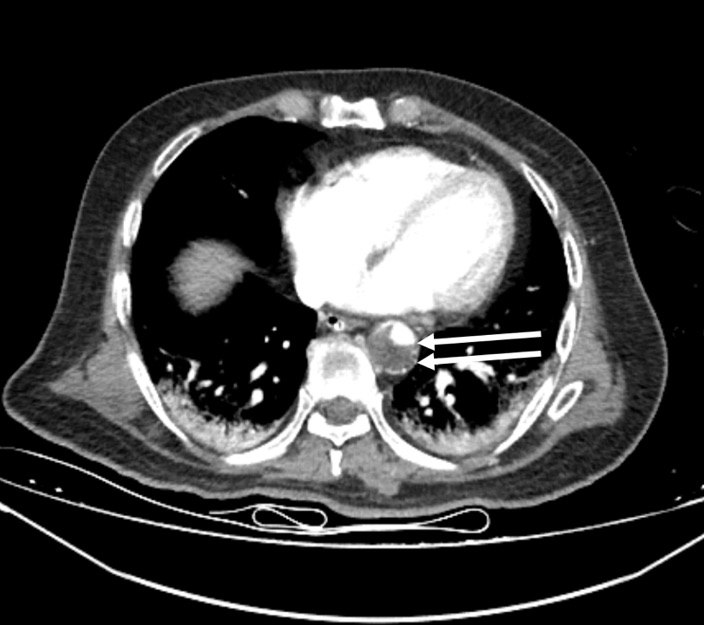
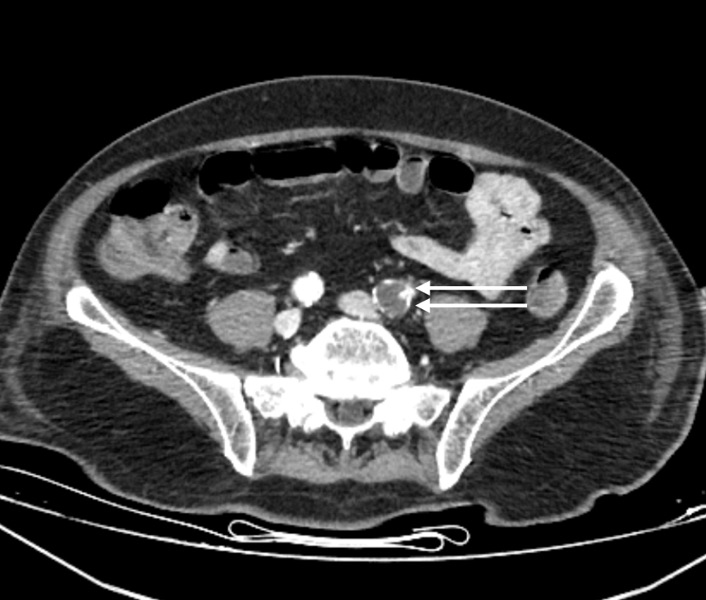
On the 13th day, the left foot was noted to be cold and ischemic, with the posterior tibial pulse not palpable. Similarly, the left hand was cold, and the radial pulse was absent. Arterial thrombosis was suspected, and Doppler ultrasonography confirmed thrombosis in the posterior tibial artery. A contrast-enhanced CT angiogram (Figure 1-3) revealed arterial thrombosis extending downward from the descending aorta, a splenic subcapsular ischemic appearance, arterial thrombosis narrowing the lumen in the left iliac and femoral arteries, and posterior tibial artery occlusion in the left leg. The patient was managed medically without surgical intervention, pentoxifylline 1500 mg/24 h infusion, dipyridamole 75 mg 1x1 tablet was started, and enoxaparin was increased to 60 mg twice daily.
On the 20th day, fresh blood was observed in the lower GI tract, and due to hemoglobin levels dropping below 70 g/dL, 2 units of erythrocyte suspension were transfused. Gastroenterology recommended monitoring for GI bleeding. Anticoagulant therapy was reduced to a prophylactic dose. Orthopedic consultation reported absent pulses in the left lower extremities and recommended amputation when a demarcation line developed.
On the 21st day, the patient was extubated and monitored with 5 L/min nasal oxygen; GCS was E4M6V4. On the 23rd day, the case was re-discussed in the council. The following decisions were made:
- No thrombectomy or similar interventional procedure for the left foot
- Continue enoxaparin 2×0.6 ml as anticoagulant therapy
- Delay contrast-enhanced CT for thrombus control due to high creatinine; perform non-contrast CT instead
- Non-contrast brain CT and neurology consultation for altered consciousness
- Wait for the demarcation line before possible amputation of the left foot
On the 27th day, hemoglobin levels dropped again, necessitating the transfusion of 1 unit of erythrocyte suspension. On the 30th day, the patient’s general condition deteriorated, requiring intubation and mechanical ventilation. The patient suffered cardiac arrest and passed away the same day.
Discussion
Some studies from China reported that 40% of hospitalized patients with COVID-19 were at high risk of VTE. Myocardial injury in COVID-19 is associated with severe disease. DIC is common in COVID-19. Regular laboratory monitoring of platelet count, PT, D-dimer, and fibrinogen in patients with COVID-19 is important to diagnose worsening coagulopathy (7).
Malas et al., in a review of 8,271 COVID-19 patients, found ICU VTE incidence at 21%, with rates up to 31%. ICU VTE rates are higher than typical for infections. The deep venous thrombosis (DVT) was 28%, the pulmonary embolism (PE) 19%, and the arterial thromboembolism (ATE) 5%. Thromboembolism raised mortality odds by 74% (8).
COVID-19 mainly affects the respiratory system but can involve other systems, notably the vascular system, which contributes significantly to morbidity and mortality. This increased thromboembolism risk arises from systemic inflammation, endothelial injury caused by the virus attaching to angiotensin-2 receptors, and viral replication, leading to prothrombotic endothelial dysfunction. To address this, the International Society of Thrombosis and Hemostasis (ISTH) interim guidance recommends considering prophylactic low molecular weight heparin (LMWH) for all hospitalized COVID-19 patients without contraindications. Similarly, the American Society of Hematology advises pharmacologic thromboprophylaxis with LMWH or fondaparinux for all hospitalized COVID-19 patients (8).
We recommend implementing standardized thromboprophylaxis for all COVID-19 patients without contraindications. Some authors advocate for higher anticoagulation targets in severely ill patients when standard dose thromboprophylaxis fails to prevent life-threatening thromboembolic complications (8).
Bellosta et al., in their study, reported that the number of patients with Acute Limb Ischemia (ALI) significantly increased in 2020 compared to the same period in 2019, and the high rate of clinical and technical failure was consistent with the presence of a hypercoagulable state triggered by COVID-19 infection (9).
Özen et al., in their study, evaluated 40 COVID-19 patients with ALI, and they mentioned that high levels of D-dimer may have predictive value for the occurrence of arterial thromboembolic events, and in some selected comorbid patients, medical follow-up may be superior to surgery (10).
Yıldız et al. in their study presented a 67-year-old male with upper limb and mesenteric ischemia and mentioned that clinicians should be alert that aortic thrombus may also be a source of embolism in COVID-19 patients, and thoracoabdominal CT angiography should be considered in routine evaluation (11). Bilge et al. in their case report, mentioned that peripheral nerve block may be a better method than general anesthesia and it has important advantages in terms of respiratory and hemodynamic (12).
Hang et al., in their study of 183 enrolled COVID-19 pneumonia patients, showed that the non-survivors had significantly higher D-dimer and fibrin degradation product (FDP) levels and longer ProThrombin than survivors on admission. DIC appeared in most of the deaths. At the late stages of COVID-19 pneumonia, levels of fibrin-related markers (D-dimer and FDP) were moderately or markedly elevated in all deaths, which suggested a common coagulation activation and secondary hyperfibrinolysis condition in these patients (13).
Conclusion
COVID-19 creates a pro-thrombotic environment, and thromboembolism is commonly observed. These complications are more often in the form of VTE. Arterial thrombosis or embolism is less common. The aortic thrombosis case we present is an example of a rare arterial thrombosis case. Thromboembolic complications can also occur in patients receiving prophylaxis with LMWH or unfractionated heparin. Therefore, unless contraindicated, higher doses than those normally used should be preferred. In select cases, adding antiplatelet therapy to anticoagulation may be considered, although further evidence is needed.
Ethical approval
Written informed consent was obtained from the participant.
Author contribution
Study conception and design: KA, MET; data collection: KA, MET; analysis and interpretation of results: KA, MET; draft manuscript preparation: KA, MET. The author(s) reviewed the results and approved the final version of the article.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Jin YH, Cai L, Cheng ZS, et al. A rapid advice guideline for the diagnosis and treatment of 2019 novel coronavirus (2019-nCoV) infected pneumonia (standard version). Mil Med Res. 2020;7:4. https://doi.org/10.1186/s40779-020-0233-6
- Lu R, Zhao X, Li J, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565-74. https://doi.org/10.1016/S0140-6736(20)30251-8
- Zheng F, Tang W, Li H, Huang YX, Xie YL, Zhou ZG. Clinical characteristics of 161 cases of corona virus disease 2019 (COVID-19) in Changsha. Eur Rev Med Pharmacol Sci. 2020;24:3404-10. https://doi.org/10.26355/eurrev_202003_20711
- Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382:1708-20. https://doi.org/10.1056/NEJMoa2002032
- World Health Organization (WHO). COVID-19 epidemiological update - 6 November 2024. Available at: https://www.who.int/publications/m/item/covid-19-epidemiological-update-edition-173
- Al-Ani F, Chehade S, Lazo-Langner A. Thrombosis risk associated with COVID-19 infection. A scoping review. Thromb Res. 2020;192:152-60. https://doi.org/10.1016/j.thromres.2020.05.039
- Bikdeli B, Madhavan MV, Jimenez D, et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75:2950-73. https://doi.org/10.1016/j.jacc.2020.04.031
- Malas MB, Naazie IN, Elsayed N, Mathlouthi A, Marmor R, Clary B. Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: a systematic review and meta-analysis. EClinicalMedicine. 2020;29:100639. https://doi.org/10.1016/j.eclinm.2020.100639
- Bellosta R, Luzzani L, Natalini G, et al. Acute limb ischemia in patients with COVID-19 pneumonia. J Vasc Surg. 2020;72:1864-72. https://doi.org/10.1016/j.jvs.2020.04.483
- Özen A, Yiğit G, Yıldırım A, Gül EB, Yılmaz M, İşcan HZ. Our clinical experience in the management of COVID-19-related arterial thrombosis with acute limb ischemia. Cardiovasc Surg Int. 2024;11:33-41. https://doi.org/10.5606/e-cvsi.2024.1609
- Yıldız İ, Hamideyin Ş, Duman Z, Güngördü F, Korkmaz M. A rare case of multiple thrombosis associated with COVID-19 pneumonia. Turk Kardiyol Dern Ars. 2022;50:466-9. https://doi.org/10.5543/tkda.2022.22379
- Bilge A, Karasoy İ, Neziroğlu E, Güner Y. Upper extremity arterial thromboembolism in a patient with severe COVID-19 pneumonia: a case report. Jt Dis Relat Surg. 2021;32:551-5. https://doi.org/10.52312/jdrs.2021.82766
- Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844-7. https://doi.org/10.1111/jth.14768
Copyright and license
Copyright © 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.