Abstract
Objective: During the pandemic, molnupiravir and favipiravir treatments have been applied at different stages of the treatment of COVID-19 patients. However, their effectiveness in COVID-19 patients admitted to the ICU is not clear.
Methods: Patients who were admitted to the ICU between March 1, 2022, and December 31, 2023 and managed according to the guidelines of the Scientific Committee of the Turkish Ministry of Health were included.
Results: Data of 152 patients who met the inclusion criteria for the study were evaluated. Patients were divided into two groups according to the treatment modality applied. When both groups were compared in terms of median hospital stay; It was found to be statistically significant (p=0.025) in Group F and 16 (11) days in Group M. When respiratory support was examined in the entire cohort; the percentage of invasive mechanical ventilation application was found to be significant as 40 (60.5) in Group F and 61 (80.3) in Group M (p=0.008). Similarly, the number of days of invasive mechanical ventilation was found to be significant as 9 (14) in Group F and 12 (8) in Group M (p=0.010). On the other hand, when evaluated in terms of hospital mortality percentage; it was found to be 42 (55.3) in Group F and 39 (51.3) in Group M and no statistically significant difference was found (p=0.626).
Conclusion: In our study, no difference was found in terms of mortality rate between molnupiravir and favipiravir, two antiviral drugs used in COVID-19 treatment. However, further studies are needed to clarify this issue.
Keywords: intensive care unit, COVID-19, anti-viral drugs, mortality rate
Introduction
Coronavirus (CoV) is a member of a large family of viruses that can cause a wide range of illnesses, from mild, self-limiting infections like the common cold to more severe conditions such as Middle East Respiratory Syndrome (MERS) and Severe Acute Respiratory Syndrome (SARS) (1). The novel coronavirus 2019 (COVID-19) was declared a “Pandemic” by the World Health Organization (WHO) on March 9, 2020 (2). Unfortunately, since then, the COVID-19 outbreak has continued to cause significant morbidity and mortality both in our country and worldwide. According to WHO data as of January 19, 2025, there have been 777,335,228 confirmed cases globally, with 7,084,023 deaths (3). In our country, according to the Ministry of Health’s data as of April 7, 2023, the total number of COVID-19 cases is reported to be 17,232,066, with 102,174 deaths (4). Approximately 20% of patients diagnosed with COVID-19 require hospitalization, and about 10% of these patients need intensive care (5). Intensive care units (ICUs) play a critical role in the treatment of COVID-19 patients, with mortality rates among critically ill COVID-19 patients in ICUs being as high as 43% (5).
To combat this deadly situation, the medical community continues to develop and use effective antiviral agents, as has been done in other viral outbreaks. Among the most well-known antiviral agents are remdesivir, favipiravir, lopinavir, and oseltamivir (6). Favipiravir, an oral antiviral drug, gained popularity during the first wave of COVID-19 (7). However, this drug had to enter clinical use without approval from the U.S. Food and Drug Administration (6). In our country, favipiravir has been included in the Ministry of Health’s COVID-19 treatment guidelines (8). Favipiravir is an RNA polymerase inhibitor with activity against a range of RNA viruses (9). It was initially licensed for the treatment of influenza viruses. Later, its inhibitory effects against COVID-19 were first identified in in vitro studies (10).
Subsequently, molnupiravir was added to these antiviral agents (11). Due to its significant inhibitory effects against Venezuelan, Eastern, and Western equine encephalitis viruses in cell cultures, it has been used for the treatment of influenza viruses and encephalitic alphaviruses. Initially developed for the treatment of influenza viral infections, this drug was later found to exhibit strong antiviral activity against SARS-CoV-2, leading to further research in this area (12). A reduction in viral RNA load has been demonstrated in in vivo studies, and the drug has been included in WHO treatment guidelines for COVID-19 (6). In our country, the Ministry of Health updated the COVID-19 adult patient guidelines on February 12, 2022, adding molnupiravir to the treatment protocols for patients over 65 years of age, those in risk groups, and hospitalized patients (8).
Both molnupiravir and favipiravir may reduce disease transmission and progression to severe illness (9). However, the safety and efficacy of these drugs in critically ill COVID-19 patients monitored in the ICU remain unclear (13). The aim of this study is to investigate the impact of molnupiravir and favipiravir use on treatment processes, clinical characteristics, and mortality rates in critically ill COVID-19 patients monitored in the ICU.
Materials and Methods
Study design and participants
This study was conducted as a retrospective analysis after obtaining approval from the University Ethics Committee (Approval ID: 2022/38-11). It involved a cohort of patients monitored in a tertiary intensive care unit (ICU) designated for COVID-19 patients between March 1, 2022, and December 31, 2023. Electronic medical records and laboratory data were obtained from the hospital database. Due to the retrospective nature of the study, written informed consent was waived.
Inclusion criteria
Patients admitted to the ICU who tested positive for COVID-19 via polymerase chain reaction (PCR), were over 18 years of age, and received either favipiravir or molnupiravir treatment were included in the study.
Exclusion criteria
Patients with negative PCR test results, those under 18 years of age, pregnant or lactating women, patients with incomplete health records, and those who did not receive favipiravir or molnupiravir treatment were excluded from the study.
Treatment protocol
In the tertiary ICU where the study was conducted, the treatment of COVID-19 patients followed the antiviral drug protocols outlined in the treatment guidelines of the Ministry of Health. According to these guidelines, favipiravir was the primary antiviral treatment used for COVID-19 in our country. Subsequently, the Ministry of Health updated the COVID-19 adult patient guidelines on February 12, 2022, adding molnupiravir to the treatment protocol for patients over 65 years of age, those in risk groups, hospitalized patients, and those monitored in the ICU (8).
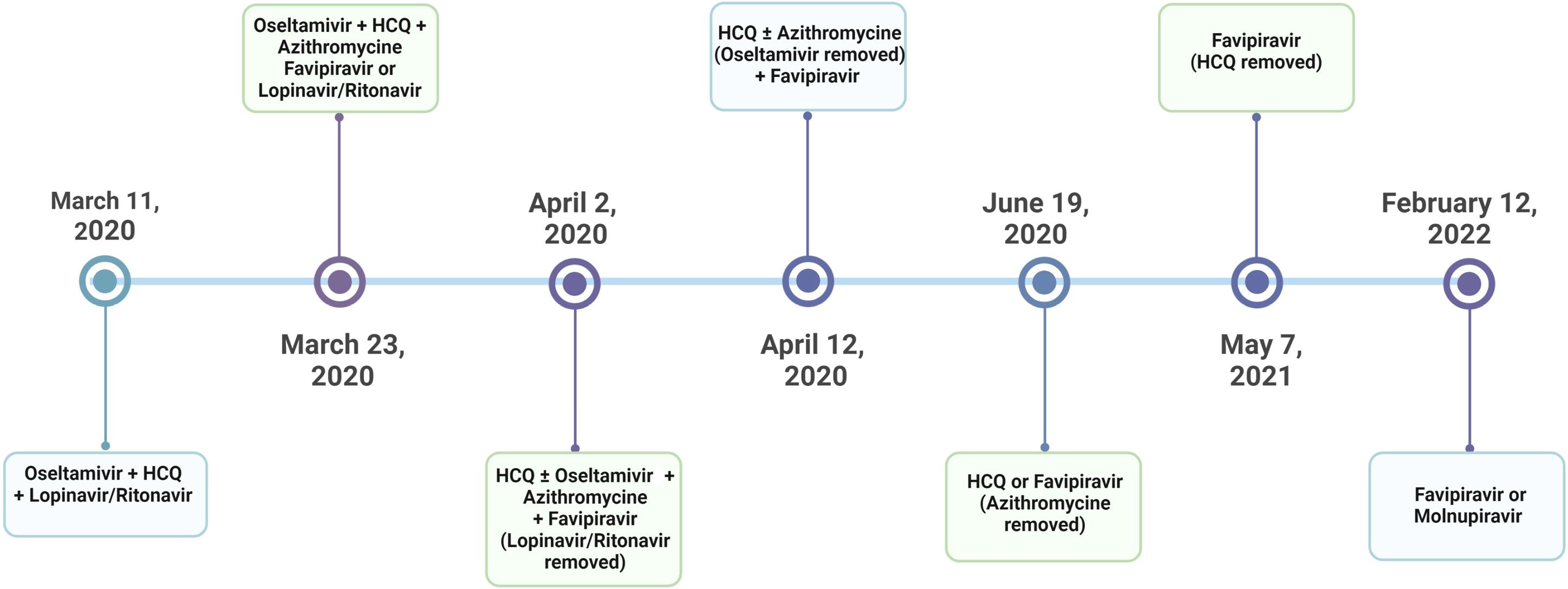
In the ICU, treatment dosages were administered according to the doses specified in the Ministry of Health guidelines. Favipiravir treatment was administered at 1600 mg twice daily on the first day, followed by 600 mg twice daily for the next 4 days. In rare cases, the treatment duration was extended to 10 days. Similarly, after the addition of molnupiravir to the guidelines, the drug was administered at 800 mg twice daily for a total of 5 days. Patients who completed both treatment protocols at the specified doses and durations were included in our study. Below is a chronological summary of the Ministry of Health COVID-19 treatment guidelines and the sequence of drug administration (14) (Figure 1).
Patient characteristics
We evaluated 205 patients (the total number of patients before matching) with critical illness due to COVID-19 and who were monitored in the intensive care unit during the specified period. Of these patients, 177 patients (the number of patients in both groups before matching) received either Favipiravir or Molnupiravir treatment. When these patients were examined, it was determined that 96 patients in the Favipiravir group and 81 patients (the number of patients in both groups before matching) met the study inclusion criteria. Patients receiving molnupiravir and favipiravir were matched according to baseline characteristics such as age, gender, body mass index, smoking status, hospitalization duration, laboratory values, respiratory support therapies (such as non-invasive or invasive mechanical ventilation), comorbidities, and intensive care unit scores. Patients with dissimilar characteristics were excluded from the analysis. Unmatched patients were not analyzed separately. Propensity Score Matching (PSM) was used to create balanced groups, and this matching was performed using a 1:1 nearest neighbor matching algorithm. Of these patients, 76 (50%) received favipiravir, and 76 (50%) received molnupiravir. Patients were divided into two groups according to the treatment modality: Favipiravir Group (Group-F) and Molnupiravir Group (Group-M).
Data collection
The following data were scanned from the electronic hospital database for each patient: age, gender, body mass index, Charlson comorbidity index (CCI), Acute Physiology and Chronic Health Evaluation (APACHE) II, and SOFA scores at ICU admission. CT-SS, arterial blood gas analysis (arterial partial pressure of oxygen (PaO2); arterial partial pressure of carbon dioxide (PaCO2); FiO2; PaO2/FiO2 ratio; bicarbonate; SpO2 (peripheral oxygen saturation)), laboratory data including hemogram parameters, C-reactive protein (CRP), procalcitonin, lactate dehydrogenase (LDH), alanine aminotransferase (ALT), aspartate aminotransferase (AST), D-dimer, serum creatinine (sCr), total bilirubin, ferritin, high-sensitive (HS) troponin I, length of hospital stay, length of intensive care unit stay, and intensive care unit and hospital mortality (Table 1).
|
BMI: Body Mass index, ABG: Arterial blood gas, ACI: Acute cardiac injury, ARDS: Acute respiratory distress syndrome, AFIB: Atrial fibrillation, AKI: Acute kidney injury, ALT: Alanine transferase, AST: Aspartate transferase, BUN: Blood urea nitrogen, CAD: Coronary artery disease, CCI: Charlson comorbidity index, CKD: Chronic kidney disease, CRP: C-reactive protein, COPD: Chronic obstructive pulmonary disease, CVD: Cerebrovascular disease; DM: Diabetes mellitus, DVT: Deep vein thrombosis, ECMO: Extracorporeal membrane oxygenation, HT: hypertension, IQR: Interquartile range, LDH: Lactate dehydrogenase, MV: Mechanical ventilation, N/A: Not applicable, PTE: Pulmonary thromboembolism, SOFA: Sequential organ failure assessment, RRT: Renal replacement therapy, VAP: Ventilator-associated pneumonia, WBC: White blood cell. *Fisher-exact test was used. |
||||
| Table 1. Characteristics of patients who received molnupiravir versus favipiravir | ||||
|
(n: 152) |
(n: 76) |
(n: 76) |
|
|
| Age mean±sd |
|
|
|
|
| Gender (female) n (%) |
|
|
|
|
| BMI median (IQR) |
|
|
|
|
| Ex-Smoker* n (%) |
|
|
|
|
| Smoker 2* n (%) |
|
|
|
|
| Hospitalization duration days median (IQR) |
|
|
|
|
| Comorbidities, n (%) | ||||
| HT |
|
|
|
|
| DM |
|
|
|
|
| CAD |
|
|
|
|
| COPD |
|
|
|
|
| CKD (*) |
|
|
|
|
| RRT (*) |
|
|
|
|
| AFIB |
|
|
|
|
| Liver Cirrhosis |
|
|
|
|
| Dyslipidemia |
|
|
|
|
| DVT |
|
|
|
|
| PTE |
|
|
|
|
| CVD (*) |
|
|
|
|
| Neurodegenerative Disorder |
|
|
|
|
| Parkinson Disease |
|
|
|
|
| Malignancy (*) |
|
|
|
|
| Scores median (IQR) | ||||
| APACHE II |
|
|
|
|
| SOFA |
|
|
|
|
| CCI |
|
|
|
|
| Laboratory findings median (IQR) | ||||
| Creatinine (mg/dl) |
|
|
|
|
| BUN (mg/dl) |
|
|
|
|
| AST U/L |
|
|
|
|
| ALT U/L |
|
|
|
|
| LDH U/L |
|
|
|
|
| CRP mg/L |
|
|
|
|
| Ferritine µg/L |
|
|
|
|
| D-Dimer μ/mL |
|
|
|
|
| BLB* |
|
|
|
|
| WBC 10*3/uL |
|
|
|
|
| Lymphocyte 10*3/uL |
|
|
|
|
| Hemoglobin g/dl |
|
|
|
|
| Platelet 10*3/uL |
|
|
|
|
| Platokrit % |
|
|
|
|
| ABG analysis median (IQR) | ||||
| pH |
|
|
|
|
| Lactate mmol/L |
|
|
|
|
| pCO2 mmHg |
|
|
|
|
| pO2 mmHg |
|
|
|
|
| Bicarbonate mmol/L |
|
|
|
|
| FiO2 median (IQR) |
|
|
|
|
| ICUP/F* median (IQR) |
|
|
|
|
| ICUvasopressor* median (IQR) |
|
|
|
|
| Airway access, n (%) | ||||
| Tracheostomy |
|
|
|
|
| Respiratory Support, n (%) | ||||
| High-Flow Nasal Cannula |
|
|
|
|
| Noninvasive MV |
|
|
|
|
| Invasive MV |
|
|
|
|
| Respiratory Support duration by modalities (day) median (IQR) | ||||
| High-Flow Nasal Cannula |
|
|
|
|
| Noninvasive MV |
|
|
|
|
| Invasive MV |
|
|
|
|
| ECMO Treatment, n (%) |
|
|
|
|
| Positive Inotropic Therapy, n (%) |
|
|
|
|
| The time diagnosis in hospital (days median) (IQR) |
|
|
|
|
| Pre-ICU Stay Duration (days median) (IQR) |
|
|
|
|
| Complication, n (%) | ||||
| VAP |
|
|
|
|
| ARDS |
|
|
|
|
| AKI |
|
|
|
|
| ACI |
|
|
|
|
| Septic Shock |
|
|
|
|
| Mortality, n (%) |
|
|
|
|
Statistical analysis
All analyses were performed using the Statistical Package for Social Sciences (SPSS) version 24.0. Propensity score matching (PSM) was employed to create balanced groups by matching patients receiving molnupiravir and favipiravir based on baseline characteristics, including age, gender, and comorbidities, using a nearest-neighbor matching algorithm with a 1:1 ratio. The Kolmogorov-Smirnov and Shapiro-Wilk tests were used to assess normality. Parametric continuous variables were compared using Student’s t-test, while non-parametric continuous variables were compared using the Mann-Whitney U test. Results are presented as mean ± standard deviation (SD) and median (interquartile range). Categorical variables were compared using the Chi-square test or Fisher’s exact test, with results presented as frequencies (%). Variables were first evaluated using univariate analysis, followed by multivariate Cox regression analysis to predict in-hospital mortality. The Kaplan-Meier test was used to evaluate factors related to survival. Statistical significance was defined as p < 0.05.
Results
Patients characteristics
We evaluated 177 patients with critical illness due to COVID-19 being followed up in the intensive care unit. Data of 152 patients who met the inclusion criteria for the study were evaluated. Of these patients, 76 (50%) were found to have received favipiravir and 76 (50%) were found to have received molnupiravir treatment. Patients were divided into two groups according to the treatment modality applied: Group Favipiravir (Group-F) and Group Molnupiravir (Group-M). No statistically significant difference was found between the two groups in terms of demographic data, laboratory results and clinical results of the patients (Table 1).
Mean age was 69.9±12.6 in Group F and 66.1±11.4 in Group M, and no statistically significant difference was found between the two groups (p=0.051). The number of female patients in Group-F was 40 (52.3%) and the number of female patients in Group-M was 38 (50%), and no statistical difference was found between them (p=0.067). When the two groups were compared in terms of the time from diagnosis to hospitalization, the median was 4 (5) days in Group F and 3 (4) days in Group M, with no statistically significant difference between the two groups (p=0.743). When both groups were compared in terms of pre-ICU stay (pre-ICU days), the median was 6 (7) days in Group F and 5 (6) in Group M, and no statistically significant difference was found between the two groups (p=0.897). The most common comorbidities were hypertension (65.1%), diabetes mellitus (35.5%) and coronary artery disease (29.6%), respectively. The APACHE II score was 20 (15.5) in the entire cohort, 22 (15) in Group F and 19 (13) in Group M (p=0.093). Median hemoglobin values were 12.9 (12) g/dl in Group F and 12.8 (12.7) g/dl in Group M, with a p-value of 0.935 (Table 1).
Treatments and outcomes
When both groups were compared in terms of median hospital stay, it was found to be statistically significant (p=0.025) in Group F and 16(11) days in Group M. When respiratory support was examined in the entire cohort, the percentage of invasive mechanical ventilation application was found to be significant as 40 (60.5) in Group F and 61 (80.3) in Group M (p=0.008). Similarly, the number of days of invasive mechanical ventilation was found to be significant as 9 (14) in Group F and 12 (8) in Group M (p=0.010). On the other hand, when evaluated in terms of hospital mortality percentage, it was found to be 42 (55.3) in Group F and 39 (51.3) in Group M, and no statistically significant difference was found (p=0.626) (Table 1).
Risk factors for hospital mortality
When Cox regression univariate analysis was performed on risk factors affecting hospital mortality, for age: HR: 1.032, 95%CI: 1.011-1.054 and p=0.003; for Charlson comorbidity index: HR: 1.149, 95%CI: 1.060-1.247 and p=0.001; for APACHE II: HR: 1.096, 95%CI: 1.065-1.127 and p<0.001; for SOFA score: HR: 1.131, 95%CI: 1.061-1.1205 and p<0.001. When multivariate analysis was performed for favipiravir, one of the antiviral agents: HR: 1.161, 95%CI: 1.018-2.517 and p=0.041; for hemoglobin: HR: 0.882, 95%CI: 0.788-0.987 and p=0.029; for D-Dimer: HR: 1.028, 95%CI: 1.008-1.048 and p=0.005; for acute kidney injury: HR: 2.420, 95%CI: 1.458-4.015 and p=0.001; for septic shock: HR: 3.410, 95%CI: 1.700-6.842 and p=0.001 (Table 2).
| CCI: Charlson comorbidity index, AKI: Acute kidney injury; HR: Hazard ratio; CI: Confidence interval, ACI: Acute cardiac injury, ARDS: Acute respiratory distress syndrome, AKI: Acute kidney injury, VAP: Ventilator-associated pneumonia, SOFA: Sequential organ failure assessment. | ||||||
| Table 2. Evaluation of factors related to in hospital mortality by Cox Regression analysis | ||||||
|
|
|
|||||
|
|
|
|
|
|
|
|
| Age |
|
|
|
|
|
|
| Female sex |
|
|
|
|
|
|
| Smoking |
|
|
|
|||
| CCI |
|
|
|
|
|
|
| APACHE II |
|
|
|
|||
| SOFA |
|
|
|
|||
| Antiviral medicine (Favipiravir) |
|
|
|
|
|
|
| pH |
|
|
|
|||
| Lactate |
|
|
|
|||
| Hemoglobin |
|
|
|
|||
| D-Dimer |
|
|
|
|||
| AKI |
|
|
|
|
|
|
| VAP |
|
|
|
|||
| ARDS |
|
|
|
|||
| ACI |
|
|
|
|||
| Septic shock |
|
|
|
|
|
|
Discussion
In this study, we investigated clinical outcomes, including mechanical ventilation duration, ICU length of stay, and mortality rates, in COVID-19 patients admitted to the ICU and treated with either favipiravir or molnupiravir. We found that patients treated with molnupiravir had significantly longer ICU stays, higher rates of invasive mechanical ventilation, and longer durations of mechanical ventilation compared to those treated with favipiravir. However, there were no statistically significant differences in ICU mortality or other clinical outcomes between the two treatment groups.
The need for safe and effective treatment options for COVID-19 patients has kept antiviral drugs at the forefront of clinical research. Antiviral agents can be used to minimize transmission and suppress the development of more virulent strains by reducing viral load (15). Molnupiravir, which acts by inhibiting RNA replicase, has been approved by infectious disease committees in many countries, including ours, for use in adults at high risk of progressing to severe COVID-19, which is associated with significant morbidity and mortality (16). Favipiravir, a purine nucleic acid analog that inhibits RNA-dependent RNA polymerase, was initially used as an anti-influenza drug in Japan and was later approved in China for the treatment of COVID-19 patients in March 2020 after Wang et al. demonstrated its in vitro efficacy against SARS-CoV-2 (17,18).
The efficacy of both drugs in COVID-19 patients remains unclear (19). In a study by Babayiğit et al. (14), no improvement in clinical parameters was observed in patients treated with favipiravir. These findings are consistent with other research indicating that the clinical recovery of patients receiving favipiravir was not superior to those receiving lopinavir/ritonavir in terms of hospital length of stay, ICU admission, or intubation rates (20). A meta-analysis on this topic also found no significant difference in mortality rates or the need for mechanical ventilation between favipiravir treatment and standard care (21). However, Guner et al. (22) reported that the addition of favipiravir to the treatment regimen reduced ICU admission and intubation rates in COVID-19 patients.
Similarly, there are various studies in the literature on patients treated with molnupiravir. In the MOVe-OUT study, molnupiravir treatment was associated with an 8% lower hospitalization rate in COVID-19 patients at high risk of progressing to severe disease (23). Another study also found that molnupiravir demonstrated favorable outcomes by reducing hospitalizations (24). A meta-analysis by Kamal et al. (25) confirmed the efficacy and safety of molnupiravir as an antiviral treatment for mild to moderate COVID-19 infections in high-risk patients.
Based on the available evidence from these clinical studies, it can be concluded that Molnupiravir is safe and effective in high-risk patients with mild to moderate COVID-19 infection when administered within the first five days of symptom onset. It is considered to delay disease progression by reducing hospitalization and/or mortality. Conversely, in the PANORAMIC study conducted in the United Kingdom (Adaptive Trial of Novel Antivirals for Early Treatment of COVID-19 in the Community), the lack of clinical benefits of Molnupiravir compared to standard care in non-hospitalized patients raised concerns. Furthermore, although the MOVe-OUT study demonstrated that Molnupiravir reduced hospitalization and mortality rates in non-hospitalized patients, the MOVe-IN study failed to show a survival advantage in the inpatient setting. However, another study found a lower risk of death among hospitalized patients treated with Molnupiravir, attributing the difference in findings between the MOVe-IN study and the present study to the inclusion of patients who initiated treatment more than five days after symptom onset.
In our study, we observed that in patients treated with Molnupiravir, the intensive care unit (ICU) length of stay, the percentage of patients requiring invasive mechanical ventilation, and the duration of ventilation were significantly higher compared to those treated with Favipiravir. However, no significant difference was detected between the two drug groups regarding ICU mortality rates. This finding may be explained by the initiation of both drugs only after hospitalization and/or ICU admission. Unfortunately, COVID-19 is a disease with a high potential for both vertical and horizontal transmission. Asymptomatic individuals play a crucial role in ongoing transmission, accounting for 25% to 50% of all new infections, which supports the recommendation for universal masking. Viral shedding begins one to two days before symptom onset, with viral titers in respiratory secretions reaching peak levels in the early stages of infection and decreasing over time. Consequently, in our study, the inability to administer the drugs during the early pre-symptomatic phase may have resulted in the loss of this critical therapeutic window. In alignment with our findings, a randomized controlled trial comparing these two drugs in COVID-19 patients also failed to demonstrate an improvement in clinical outcomes.
Limitations
Our study has inherent limitations due to its retrospective nature. Additionally, there are other constraints. Firstly, this is a single-center study with a relatively small patient population. Secondly, the exact timing of symptom onset was not clearly documented for each patient. Thus, the treatment doses and durations analyzed in this study reflect those administered at the hospital or ICU level. As a result, evaluating the clinical recovery process in cases where Favipiravir or Molnupiravir was administered during the early symptomatic phase remains insufficient. On the other hand, we think that our study is important in that it reflects the treatment processes of COVID-19 patients treated with antiviral drugs in the COVID intensive care unit of a university hospital during the pandemic.
Conclusion
In our study, no difference in mortality rates was found between the two antiviral drugs, Molnupiravir and Favipiravir, used for COVID-19 treatment. However, compared to Favipiravir-treated patients, those treated with Molnupiravir had significantly longer ICU stays, higher rates of invasive mechanical ventilation, and prolonged ventilation duration. We hypothesize that this finding may be related to the delayed administration of these drugs rather than their early use in the disease course. Nevertheless, further studies are needed to validate this observation.
Ethical approval
This study has been approved by the Dokuz Eylül University Non-Interventional Research Ethics Committee (approval date: 30.11.2022, number: 2022/38-11). Written informed consent was obtained from the participants.
Author contribution
Study conception and design: ÖÖ; data collection: MÇG, NY; analysis and interpretation of results: BE; draft manuscript preparation: VH, ANG, EY. The author(s) reviewed the results and approved the final version of the article.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- V’kovski P, Kratzel A, Steiner S, Stalder H, Thiel V. Coronavirus biology and replication: implications for SARS-CoV-2. Nat Rev Microbiol. 2021;19:155-70. https://doi.org/10.1038/s41579-020-00468-6
- Attwood SW, Hill SC, Aanensen DM, Connor TR, Pybus OG. Phylogenetic and phylodynamic approaches to understanding and combating the early SARS-CoV-2 pandemic. Nat Rev Genet. 2022;23:547-62. https://doi.org/10.1038/s41576-022-00483-8
- World Health Organization (WHO). Coronavirus disease (COVID-19) pandemic. WHO; 2019. Available at: https://www.who.int/emergencies/diseases/novel-coronavirus-2019
- T.C. Sağlık Bakanlığı. Genel koronavirüs tablosu. Ankara: T.C. Sağlık Bakanlığı; 2022. Available at: https://covid19.saglik.gov.tr/TR-66935/genel-koronavirus-tablosu.html
- Öner Ö, Ergan B, Gürkök MÇ, et al. Is there a relationship between mortality rates and nutritional factors in critical ill patients with COVID-19? Turk J Intensive Care. 2023;21:74-82. https://doi.org/10.4274/tybd.galenos.2022.92905
- Mali KR, Eerike M, Raj GM, et al. Efficacy and safety of molnupiravir in COVID-19 patients: a systematic review. Ir J Med Sci. 2023;192:1665-78. https://doi.org/10.1007/s11845-022-03139-y
- Singh AK, Singh A, Singh R, Misra A. Molnupiravir in COVID-19: a systematic review of literature. Diabetes Metab Syndr. 2021;15:102329. https://doi.org/10.1016/j.dsx.2021.102329
- T.C. Sağlık Bakanlığı. Ağır pnömoni, ARDS, sepsis ve septik şok yönetimi. Ankara: T.C. Sağlık Bakanlığı; 2025. Available at: https://covid19.saglik.gov.tr/TR-66340/agir-pnomoni-ards-sepsis-ve-septik-sok-yonetimi.html
- Yasri S, Wiwanitki V. Molnupiravir, favipiravir and other antiviral drugs with proposed potentials for management of COVID-19: a concern on antioxidant aspect. Int J Biochem Mol Biol. 2022;13:1-4.
- Ghasemnejad-Berenji M, Pashapour S. Favipiravir and COVID-19: a simplified summary. Drug Res (Stuttg). 2021;71:166-70. https://doi.org/10.1055/a-1296-7935
- Tian L, Pang Z, Li M, et al. Molnupiravir and its antiviral activity against COVID-19. Front Immunol. 2022;13:855496. https://doi.org/10.3389/fimmu.2022.855496
- Agostini ML, Pruijssers AJ, Chappell JD, et al. Small-molecule antiviral β-d-N4-Hydroxycytidine inhibits a proofreading-intact coronavirus with a high genetic barrier to resistance. J Virol. 2019;93:e01348-19. https://doi.org/10.1128/JVI.01348-19
- Jean SS, Lee PI, Hsueh PR. Treatment options for COVID-19: the reality and challenges. J Microbiol Immunol Infect. 2020;53:436-43. https://doi.org/10.1016/j.jmii.2020.03.034
- Babayigit C, Kokturk N, Kul S, et al. The association of antiviral drugs with COVID-19 morbidity: the retrospective analysis of a nationwide COVID-19 cohort. Front Med (Lausanne). 2022;9:894126. https://doi.org/10.3389/fmed.2022.894126
- Panda PK, Bandyopadhyay A, Singh BC, et al. Safety and efficacy of antiviral combination therapy in symptomatic patients of Covid-19 infection - a randomised controlled trial (SEV-COVID Trial): a structured summary of a study protocol for a randomized controlled trial. Trials. 2020;21:866. https://doi.org/10.1186/s13063-020-04774-5
- Wen W, Chen C, Tang J, et al. Efficacy and safety of three new oral antiviral treatment (molnupiravir, fluvoxamine and Paxlovid) for COVID-19: a meta-analysis. Ann Med. 2022;54:516-23. https://doi.org/10.1080/07853890.2022.2034936
- Korula P, Alexander H, John JS, et al. Favipiravir for treating COVID-19. Cochrane Database Syst Rev. 2024;2:CD015219. https://doi.org/10.1002/14651858.CD015219.pub2
- WHO Solidarity Trial Consortium , Pan H, Peto R, et al. Repurposed antiviral drugs for Covid-19 - interim WHO solidarity trial results. N Engl J Med. 2021;384:497-511. https://doi.org/10.1056/NEJMoa2023184
- Eloy P, Le Grand R, Malvy D, Guedj J. Combined treatment of molnupiravir and favipiravir against SARS-CoV-2 infection: one + zero equals two? EBioMedicine. 2021;74:103663. https://doi.org/10.1016/j.ebiom.2021.103663
- Amani B, Khanijahani A, Amani B, Hashemi P. Lopinavir/ritonavir for COVID-19: a systematic review and meta-analysis. J Pharm Pharm Sci. 2021;24:246-57. https://doi.org/10.18433/jpps31668
- Özlüşen B, Kozan Ş, Akcan RE, et al. Effectiveness of favipiravir in COVID-19: a live systematic review. Eur J Clin Microbiol Infect Dis. 2021;40:2575-83. https://doi.org/10.1007/s10096-021-04307-1
- Guner AE, Surmeli A, Kural K, et al. ICU admission rates in Istanbul following the addition of favipiravir to the national COVID-19 treatment protocol. North Clin Istanb. 2021;8:119-23. https://doi.org/10.14744/nci.2021.60420
- Guan Y, Puenpatom A, Johnson MG, et al. Impact of molnupiravir treatment on patient-reported COVID-19 symptoms in the phase 3 MOVe-OUT trial: a randomized, placebo-controlled trial. Clin Infect Dis. 2023;77:1521-30. https://doi.org/10.1093/cid/ciad409
- Karniadakis I, Mazonakis N, Tsioutis C, Papadakis M, Markaki I, Spernovasilis N. Oral molnupiravir and nirmatrelvir/ritonavir for the treatment of COVID-19: a literature review with a focus on real-world evidence. Infect Dis Rep. 2023;15:662-78. https://doi.org/10.3390/idr15060061
- Kamal L, Ramadan A, Farraj S, Bahig L, Ezzat S. The pill of recovery; molnupiravir for treatment of COVID-19 patients; a systematic review. Saudi Pharm J. 2022;30:508-18. https://doi.org/10.1016/j.jsps.2022.03.002
Copyright and license
Copyright © 2026 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.